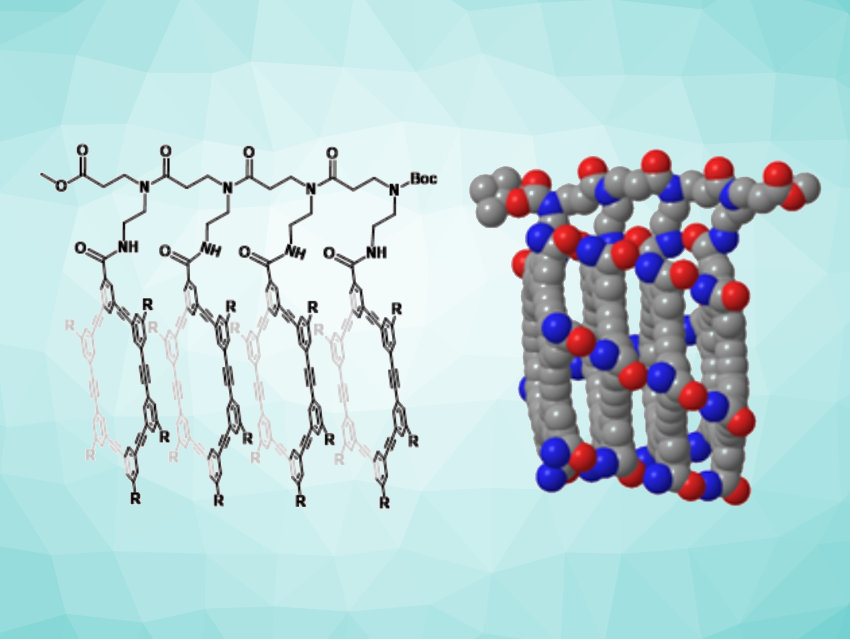
Channels and pores are important in biological systems to mediate the movement of ions and molecules across cell membranes. Synthetic tube-like structures can mimic the properties of natural channels and pores. They can be created, e.g., by using self-assembly to stack macrocyclic molecules and create nanotubes. Hexakis(m-phenylene ethynylene) macrocycles, for example, can stack to form nanotubes. However, the interactions between the macrocycles in such systems are usually dynamic, leading to varying stabilities. Creating covalent bonds between the molecules could provide improved stabilities, as well as defined tube lengths.
Rui Liu, Zhong-Lin Lu, Beijing Normal University, China, and colleagues have developed a strategy for creating molecular nanotubes with defined lengths using covalently linked hexakis(m-phenylene ethynylene) macrocycles (simplified example pictured). The team first synthesized macrocycles with two types of side chains on the periphery. One type of the side chain has a β-alanine methyl ester motif, which serves as a linker to tether the macrocycles together, and the other is a chiral dioctanyl-substituted aminopropylene glycol ether side chain, which induces chiral helical stacking and improves the solubility of the macrocyclic compounds. Then they connected the macrocycles via amide bonds using common peptide chemistry to form stacks of either two or four macrocyclic units.
The researchers found that the nanotube comprised of four macrocycles can form a channel that can transport protons or potassium ions across lipid bilayers. The channels remained open continuously for ca. 60 s, which is among the longest open durations for synthetic ion channels according to the team. Overall, the work shows that covalently tethering shape-persistent macrocyclic units together is a useful approach to building molecular nanotubes.
- Covalently Linked Hexakis(m‐Phenylene Ethynylene) Macrocycles as Molecular Nanotubes,
Cheng-Yan Wu, Shilong Su, Xi Zhang, Rui Liu, Bing Gong, Zhong-Lin Lu,
Angew. Chem. Int. Ed. 2023.
https://doi.org/10.1002/anie.202303242