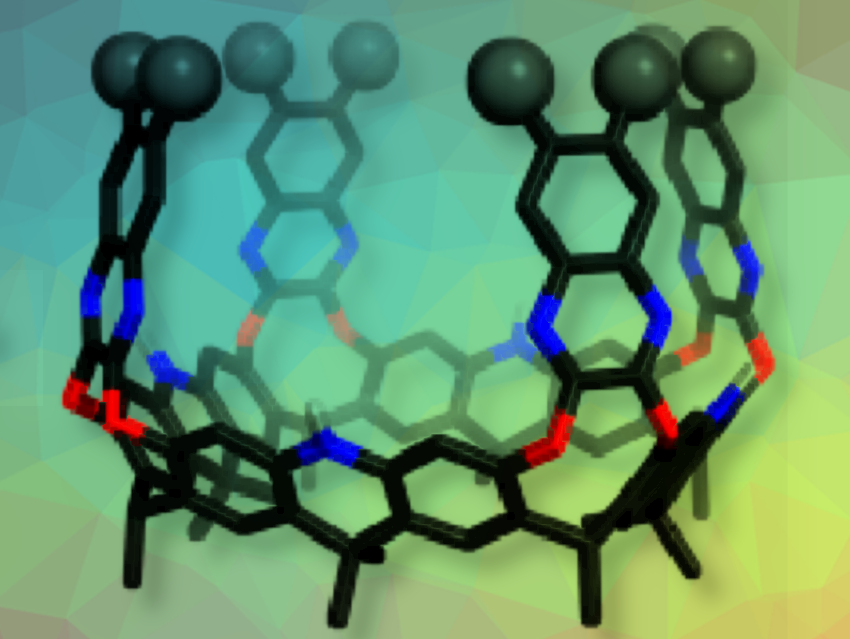
Organic molecular containers on the nanometer scale have found wide applications in the fields of sensing, purification, and catalysis. The construction of such nanocontainers requires large bowl-shaped molecules, called cavitands, the size of which has been a limiting factor for the construction of large nanocontainers. The cavitand volume is restricted by the size of the structural platform used (diameter approx. 7 Å).
Konrad Tiefenbacher and colleagues, University of Basel, Switzerland, have synthesized a very large bowl-shaped molecule named acridane[4]arene (diameter approx. 14 Å) in six steps and with high yields (38 %). The crucial macrocyclization step involves an acridane precursor and dodecanal in the presence of catalytic amounts of triflic acid.
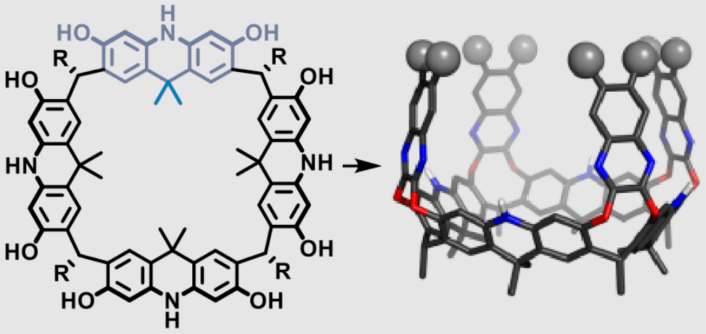
The macrocycle favors a crown conformation because of the hydrogen bond pattern of the free phenols and the preference of the R substituents for axial orientation. Nanocontainers, deep enough for C60 and C70, were synthesized by functionalizing the macrocycle with quinoxaline walls by nucleophilic aromatic substitution. The so-called megalo-cavitands have volumes of up to 814 Å3. The selective binding of fullerene C70 in the presence of C60 was demonstrated.
The researchers anticipate that that the macrocycle will be useful for the construction of a wide range of nanocontainers, including those that are water soluble and able to interact with protein surfaces.
- Megalo‐Cavitands: Synthesis of Acridane[4]arenes and Formation of Large, Deep Cavitands for Selective C70 Uptake,
Jonathan Pfeuffer-Rooschüz, Salome Heim, Alessandro Prescimone, Konrad Tiefenbacher,
Angew. Chem. Int. Ed. 2022.
https://doi.org/10.1002/anie.202209885