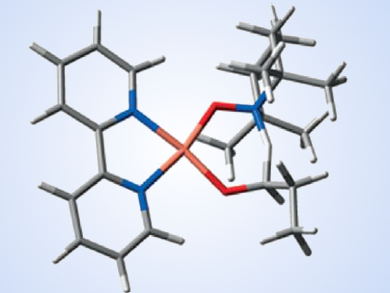
The oxidation of alcohols plays a huge role within chemical synthesis and is used on large scales in industry. Green, non-toxic oxidizing agents have proven hard to come by. However, TEMPO (2,2,6,6-tetramethylpiperidine-N-oxyl) has proven to be a popular option. This is due to its low cost and simple use alongside Cu(I) sources as a cocatalyst. The mechanism of Cu(I)/TEMPO-catalyzed aerobic oxidation has eluded chemists since its development, particularly regarding an intramolecular hydrogen-transfer step and the structure of a transition state.
Alex Szpilman, Ariel University, Israel, and Mark A. Iron, Weizmann Institute of Science, Rehovot, Israel, have studied the mechanism using density functional theory (DFT) calculations. Modeling the oxidation of three different alcohols using a Cu(I) complex with bipyridine and N-methylimidazole ligands, the team found a likely mechanism for the reaction.
The cycle is proposed to involve hydrogen transfer to the TEMPO nitrogen atom via a 6-membered transition state, which is far more energetically favorable than the alternative hydrogen transfer to the oxygen atom. Crucially, it was suggested that this oxidation through hydrogen transfer is not the rate-determining step as proposed in previous research, but rather that the oxygen activation or alcohol activation steps were more likely to be rate-determining.
Understanding the catalytic mechanism is an important prerequisite to catalyst design. This work could pave the way towards new and improved industrial oxidation catalysts, suitable for a wide range of substrates, and bearing the principles of green chemistry in mind.
- Mechanism of the Copper/TEMPO-Catalyzed Aerobic Oxidation of Alcohols,
Mark A. Iron, Alex M. Szpilman,
Chem. Eur. J. 2016.
DOI: 10.1002/chem.201604402