Single Drug to Fight Resistant Cancers
Clinicians combat the drug resistances of some cancer types by using a combination of different drugs. To make this approach more effective, Dan Gibson, The Hebrew University of Jerusalem, Israel, Gilles Gasser, Université Paris Sciences et Lettres, CNRS, France, and colleagues have designed a chemical conjugate that can simultaneously attack several cellular targets using different modes of action. Such a single-drug therapy would increase the chances of killing all cancer cells.
The most frequently clinically applied chemotherapeutic drug is cisplatin, a metal complex based on the platinum(II) ion. The drug’s mode of action is binding to the DNA in the tumor cells, where it distorts the DNA structure and ultimately triggers cell death. Other chemicals facilitate the interaction of cisplatin with DNA, and they are often combined with cisplatin for chemotherapy. The photodynamic therapy (PDT) approach, in contrast, relies on the activation of a metal complex by laser light. A reactive form of oxygen is formed, which interferes with cell metabolism, triggering cell death.
Chemotherapy and Photodynamic Therapy Combined
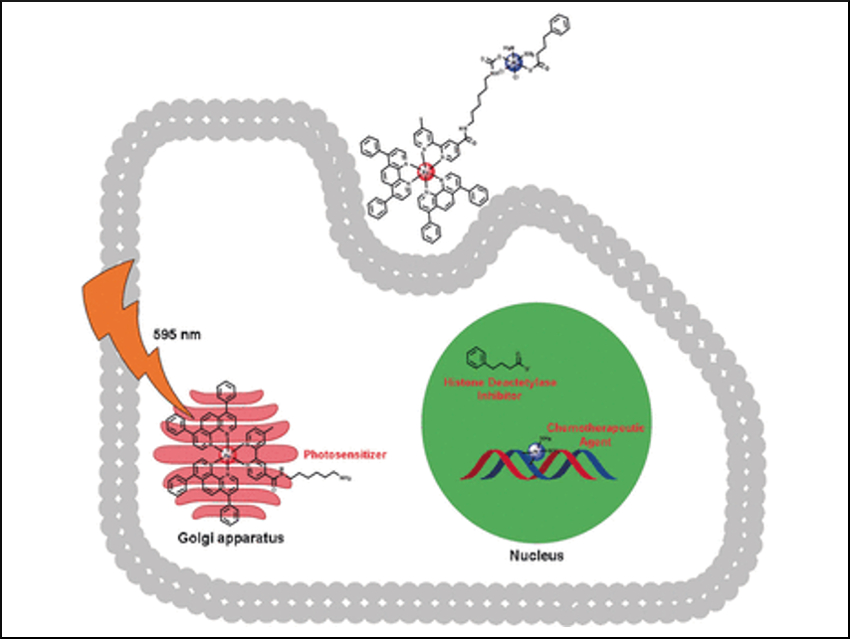
“In clinical protocols, each drug is administered separately and may not reach the tumor at the same time or at a fixed ratio,” says Gasser. The team combined cisplatin, phenylbutyrate (a chemical enhancer for cisplatin), and a PDT drug (a metal complex based on RuII), into a single compound called Ru–Pt. The idea was that the three drugs in conjunction could travel the bloodstream intact and enter their target tumor cells, which would reduce side effects and the need to adjust the dosages.
The researchers have designed the phototherapeutic Ru(II) half of Ru–Pt so that it can be excited with laser light in the deep red section of the wavelength spectrum, which penetrates deeply into biological tissue. The cisplatin- and phenylbutyrate-containing half of Ru–Pt was designed as a prodrug that is activated by components inside the cell. Both therapeutic components were attached to each other by a molecular spacer. “The correct spacer length was critical to ensure that both drug compounds will not interfere with each other, but the molecule remains small, water-soluble, and able to travel across membranes,” Gasser says.
The researchers added Ru–Pt to normal and cancer cell lines and found that Ru–Pt was significantly more efficient in killing cancer cells than the single compounds based on Ru(II) and Pt(IV). The team also reported that the irradiated samples had significantly higher tumor-killing rates, which means that the specific drug activation in tumor tissue is possible. And finally, Ru–Pt had a ten times higher efficiency for drug-resistant cell lines than the single reagents. These results demonstrate the high potential of multimodal drugs for developing more selective and effective drugs that have fewer side effects and allow for simple handling and effective cancer treatment.
- A Multi‐action and Multi‐target RuII–PtIV Conjugate Combining Cancer‐Activated Chemotherapy and Photodynamic Therapy to Overcome Drug Resistant Cancers,
Johannes Karges, Thirumal Yempala, Mickaël Tharaud, Dan Gibson, Gilles Gasser,
Angew. Chem. Int. Ed. 2020.
https://doi.org/10.1002/anie.201916400