Organic light-emitting diodes (OLEDs) are driving a new generation of vivid, thin, and efficient displays. To make efficient OLEDs that are free of rare-earth metals, researchers have been focusing on a process known as thermally activated delayed fluorescence (TADF). Good blue-emitting TADF molecules, however, have been difficult to realize.
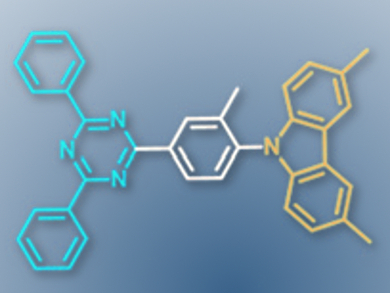
Chihaya Adachi, Kyushu University and Japan Science and Technology Agency (ERATO), both Fukuoka, Japan, and colleagues developed an advanced molecular design strategy for such TADF molecules. The team started with a basic deep-blue emitter consisting of a cyaphenine electron-accepting unit, linked to a carbazole-based electron-donating unit by a phenyl bridge (pictured).
The researchers added methyl groups on the phenyl linker to influence the twisting of the molecule. This greatly increases the TADF efficiency while barely affecting color. Adding methyl groups on the carbazole unit also increased the TADF efficiency, but led to a red-shift in emission.
The best emitter achieved an external quantum efficiency of up to 19.2 %. According to the researchers, the work could help to guide the design of new deep-blue TADF emitters.
- Controlling Singlet-Triplet Energy Splitting for Deep-Blue Thermally Activated Delayed Fluorescence Emitters,
Lin-Song Cui, Hiroko Nomura, Yan Geng, Jong Uk Kim, Hajime Nakanotani, Chihaya Adachi,
Angew. Chem. Int. Ed. 2016.
DOI: 10.1002/anie.201609459