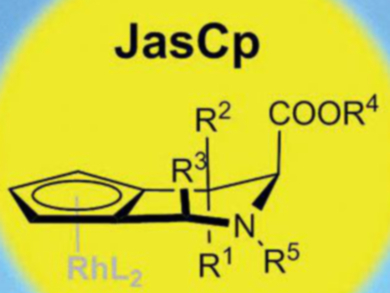
Cyclopentadienyl (Cp) ligands are widely used in versatile transition-metal-catalyzed reactions, in particular C–H bond activation. To render the corresponding transformations enantioselective, chiral ligands are needed. However, only few chiral Cp ligands have been reported so far.
Andrey P. Antonchick, Herbert Waldmann, Max Planck Institute of Molecular Physiology, Dortmund, Germany, and Technical University Dortmund, and colleagues have developed a general approach to chiral ligand discovery. The team used an enantioselective [6+3] cycloaddition of imino esters with fulvenes as the key synthesis step. The structure of the resulting chiral so-called JasCp ligands (example complex pictured) can be easily varied through a three-step synthesis on a gram scale.
The applicability of JasCp was demonstrated by the successful application in three enantioselective Rh(III)-catalyzed C–H bond activation reactions. The results suggest that this approach should enable the discovery of novel efficient enantioselective transformations using easily accessible ligands.
- General Enantioselective C–H Activation with Efficiently Tunable Cyclopentadienyl Ligands,
Zhi-Jun Jia, Christian Merten, Rajesh Gontla, Constantin G. Daniliuc, Andrey P. Antonchick, Herbert Waldmann,
Angew. Chem. Int. Ed. 2017.
DOI: 10.1002/anie.201611981