Tuberculosis (TB), caused by Mycobacterium tuberculosis (Mtb), is the leading worldwide cause of death from infectious disease. TB requires a lengthy combination treatment regimen, which has significant side-effects, resulting in poor patient compliance and the emergence of drug-resistant strains. The well-tolerated ß-lactam antibiotic class is widely prescribed for the treatment of other bacterial infections, however, Mtb has an intrinsic resistance against it.
Randall J. Basaraba, Colorado State University, Fort Collins, USA, Christian Melander, North Carolina State University, Raleigh, USA, and colleagues have identified a series of small molecules that act as antibiotic adjuvants and suppress ß-lactam resistance. These compounds possess an orthogonal spectrum of activity to that of traditional ß-lactamase inhibitors. They do not directly inhibit the ß-lactamase enzyme but act through a novel mechanism which is still to be investigated.
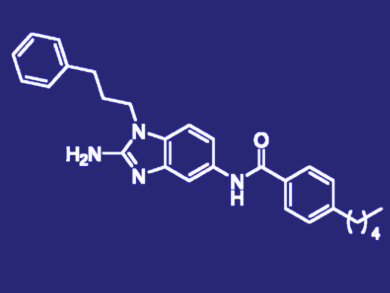
The lead compound (2-aminobenzimidazole, pictured) and its derivatives display potent activity against Mtb and suppress the resistance of several drug-resistant strains to the ß-lactam antibiotic carbenicillin. According to the researchers, this discovery could be a significant breakthrough in the identification of new therapeutic approaches for the treatment of tuberculosis.
- The Discovery of 2-Aminobenzimidazoles That Sensitize Mycobacterium smegmatis and M. tuberculosis to β-Lactam Antibiotics in a Pattern Distinct from β-Lactamase Inhibitors,
T. Vu Nguyen, Meghan S. Blackledge, Erick A. Lindsey, Bradley M. Minrovic, David F. Ackart, Albert B. Jeon, Andrés Obregón-Henao, Roberta J. Melander, Randall J. Basaraba, Christian Melander,
Angew. Chem. Int. Ed. 2017.
DOI: 10.1002/anie.201612006