Many applications in molecular electronics benefit from the controlled synthetic manipulation of graphene nanostructures to incorporate atomically precise edge structures and dopants. A bottom-up synthetic approach, using small organic building blocks, offers great potential for structural control. However, it requires highly efficient methods for the selective fusion of many aromatic rings.
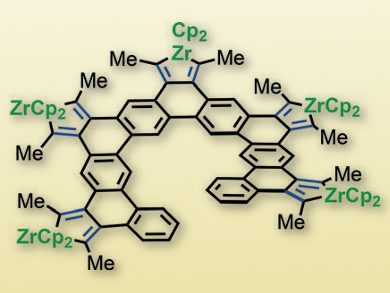
T. Don Tilley and colleagues, University of California, Berkeley, USA, have developed a strategy for the construction of well-defined graphene nanostructures. The approach is based on a quantitative intramolecular reductive cyclization of an oligo(diyne) with a low-valent zirconocene reagent to give zirconacyclopentadiene-annulated polycyclic aromatic hydrocarbons (PAH). This method produces large fused-ring systems (example pictured) from non-fused precursors, which are generally accessible using well-developed aryl-aryl couplings.
Importantly, the resulting fused-ring systems contain synthetically versatile zirconacyclopentadiene units, which might undergo well-established transformations to a wide range of functionality, including dienes, heterocycles, and more extended π-systems. Such cyclization reactions could help address challenges associated with the bottom-up construction of graphene nanostructures with tailored properties.
- Zirconacyclopentadiene-Annulated Polycyclic Aromatic Hydrocarbons,
Gavin R. Kiel, Micah S. Ziegler, T. Don Tilley,
Angew. Chem. Int. Ed. 2017.
DOI: 10.1002/anie.201700818