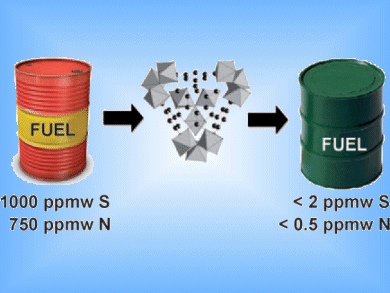
Diesel and gasoline are blends of aromatic and aliphatic compounds that contain significant levels of heterocyclic aromatic contaminants. Nitrogen compounds form NOx exhaust gases which contribute to smog while the sulfur equivalent, SOx, contributes to acid rain. Hydrodesulfurization catalysts can produce fuels with sulfur levels down to 5 ppmw (parts per million by weight), but only in the absence of nitrogen compounds.
Dirk E. De Vos and co-workers, Katholieke Universiteit Leuven, Belgium, have found that metal-organic frameworks (MOFs) can be used to remove nitrogen compounds from fuel feeds. Good uptake of a range of aromatic nitrogen compounds was seen with the iron-based MOF, MIL-11, which contains trimers of metal octahedra with Lewis acid sites.
This provides a cheap, non-toxic, and biodegradable material that can be regenerated with ease and could be used to remove nitrogen compounds before they poison hydrodesulfurization catalysts and pollute the environment.
- Selective Removal of N-Heterocyclic Aromatic Contaminants from Fuels by Lewis Acidic Metal–Organic Frameworks
M. Maes, M. Trekels, M. Boulhout, S. Schouteden, F. Vermoortele et al.,
Angew. Chem. Int. Ed. 2011.
DOI: 10.1002/anie.201100050