The lack of selectivity of current anticancer drugs results in ineffective therapy and systemic toxicity. For solid tumors, selectivity is more difficult because these tumors reside in an environment with a heterogeneous cell population that consists of both malignant and healthy cells.
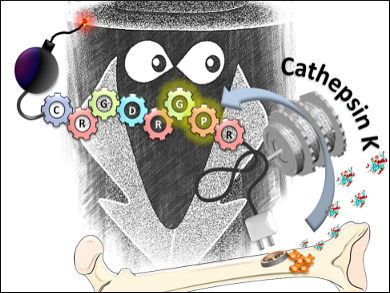
Alejandro Baeza, María Vallet-Regi, and colleagues, Centro de Investigacion Biomedica en Red de Bioingeniera, Biomateriales y Nanomedicina (CIBER-BBN), Madrid, Spain, have created a double sequential encrypted targeting system and demonstrated its applicability to bone cancer. The targeting system consists of an alendronate (ALN) molecule, which interacts with the hydroxyapatite mineral in bones. ALN is attached to a polyethylene glycol (PEG) linker. The other side of the PEG linker is then attached to a peptide sequence containing (1) the amino acid sequence Arg-Gly-Asp (RGD), (2) a PEG-linked fluorescent tag, and (3) the cleavable sequence Arg-Pro-Gly (RPG). The RGD part is a ligand for integrins, which are over-expressed in many tumor cells and the RPG sequence is cleavable by cathepsin K – an enzyme present in high amounts in bone tissues with bone tumors.
After in vitro characterization using fluorescence microscopy and mass spectrometry, the team evaluated the performance of their targeting system to human osteosarcoma (HOS) cells. Their targeting system did not bind to HOS cells without cathepsin K cleavage, showing that the ALN-PEG group hides the RGD, thereby preventing unwanted interactions. After cathepsin K cleavage, the fluorescent-tagged RGD is then internalized by HOS cells.
The researchers believe that this system may be applicable to other tumors and can be linked to different drug carriers. As such, this system presents a proof-of-concept towards synergistic targeting moieties leading to more selective drug delivery.
- Double Sequential Encrypted Targeting Sequence: A New Concept for Bone Cancer Treatment,
Gonzalo Villaverde, Valentina Nairi, Alejandro Baeza, María Vallet-Regí,
Chem. Eur. J. 2017.
DOI: 10.1002/chem.201605947