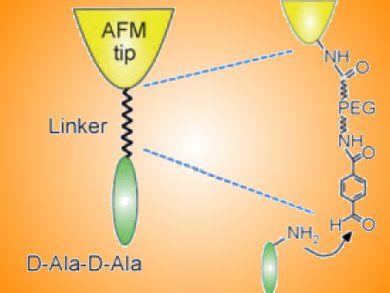
The ability of peptides to bind to surfaces is currently being explored in various fields of nanotechnology, in particular biomimetic hybrid materials. Understanding how the amino acid residues in peptides bind with inorganic surfaces could lead to new ways of functionalizing surfaces and new materials.
Jessem Landoulsi and Vincent Dupres have used atomic force microscopy (AFM) to study the binding of a single peptide to a stainless steel surface. The peptide was immobilized on the AFM tip and the steel surface prepared so that an iron oxide layer formed and remained stable. The average force required to detach the peptide from the surface was in the range of 50—300 pN. This cannot be attributed to covalent bonds; rather it implies that binding occurs via a combination of van der Waals and hydrogen bonds.
- Probing Peptide–Inorganic Surface Interaction at the Single Molecule Level Using Force Spectroscopy
J. Landoulsi, V. Dupres,
ChemPhysChem 2011.
DOI: 10.1002/cphc.201100007

this research article is opening a new area for peptide chemistry. It must absorb the world’s attention. Good.