Protein Mass Spectrometry
Electrospray ionization (ESI) is a critical step in the application of mass spectrometry. The technique can easily form intact, gaseous, multiply charged protein ions that are then subject to the rapid and sensitive analytical technique. Scientists have two models to help them explain why ESI works so well with proteins. The two models are essentially mutually exclusive, which means that while neither might be the correct explanation, they cannot both be right.
The first model is the charge residue model (CRM) and the second is the ion evaporation model (IEM). Both involve neutral molecules evaporating from a charged droplet and then splitting into fragments that show up in the mass spectrum, allowing information to be gleaned about the structure of the proteins. In the journal Angewandte Chemie, Australian researchers explain that in the CRM, a charged droplet is formed by sequential droplet evaporation and Coulombic fission events. This charged droplet carries with it a single analyte ion, which loses neutral solvent molecules until it is dry and ready to produce its mass spectrum. In the other model, the IEM, it is thought that the electric field on the droplet surface is enough to eject an analyte ion. There is competing evidence for both, the CRM and the IEM.
Highly Charged Protein Ions as Extremely Strong Acids
William A. Donald, University of New South Wales, Sydney, Australia, and colleagues have investigated the basicity of two protein ions, highly protonated cytochrome c (cyt c) and myoglobin (myo). They used tandem mass spectrometry (MS/MS), ion–molecule reactions (IMRs), and theoretical calculations in an effort to unfold the story of protein mass spectrometry and help them decide whether the CRM or the IEM is the most appropriate description of the electrospray process in mass spectrometry.
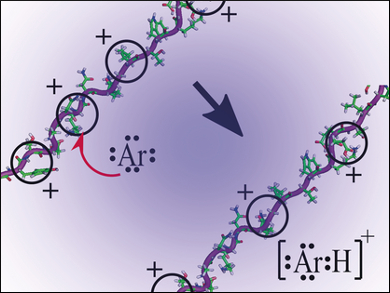
The team has shown rather surprisingly that the highly charged protein ions cyt c and myo can actually protonate non-polar molecules and even inert gases, including argon, oxygen, and nitrogen in thermal ion–molecule reactions. Indeed, the team describes these protonated proteins as the “most acidic organic species to be isolated and detected”.
The team’s theoretical work suggests that both cyt c and myo ions in the highly protonated state should readily lose a protein to vacuum conditions, once at least every third amino acid residue is protonated. In their experiments, highly protonated protein ions were formed in which every fourth residue is protonated on average. The theory hints that it might be possible to use a lower pressure ion source in ESI to reduce the proton transfer to oxygen and nitrogen or noble gases used as the atmosphere for the analytical system. “The extent of protein charging in ESI can be limited by proton transfer reactions with atmospheric gases,” the team reports. This discovery thus hints at a way to make ESI-MS work more effectively for protein studies, as well as providing a new clue as to whether CRM or IEM is the correct model to explain the ESI process.
Evidence for a Chain-Ejection Model (CEM)
The team explains: “Departing neutral solvent molecules should readily deprotonate such highly acidic protein ions near the moment of ion formation.” This implies that the protonated proteins should not survive the ESI process if the CRM is correct. Instead, it is more likely that the fairly novel chain-ejection model (CEM) – which is very similar to IEM – might come into play in these systems. In this model, protein ions are ejected from highly charged ionic droplets and competition for charge between the protein ion and highly charged ionic droplets during protein chain ejection depends on whether basicity is lowered relative to the neutral solvent molecules. Indeed, the team says, “the protein ions are more than 375 kilojoules per mole less basic than the least volatile components of ESI solutions,” this suggests that “The chain ejection model (CEM) is more likely.”
Ultimately, this finding means that ESI-MS can be fine tuned based on this explanation of the ESI process. “It is anticipated that ESI in an inert, reduced pressure atmosphere will be useful for increasing ion charging even further to maximize the performance of many tandem mass spectrometry experiments,” the team concludes.
- Highly Charged Protein Ions: The Strongest Organic Acids to Date,
Muhammad A. Zenaidee, Michael G. Leeming, Fangtong Zhang, Toby T. Funston, William A. Donald,
Angew. Chem. Int. Ed. 2017.
DOI: 10.1002/anie.201702781