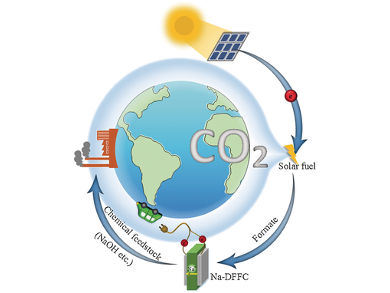
Alkaline sodium-ion conducting direct liquid fuel cells convert chemical energy directly into electricity with high energy-conversion efficiency and no emissions. However, current technology requires addition of base to the fuel solution to achieve an alkali environment with adequate charge carrier properties.
Yinshi Li, Xi’an Jiaotong University, Shaanxi, China, and colleagues have developed a sodium-ion conducting direct formate fuel cell (Na-DFFC), which converts the chemical energy stored in formate into electricity without the need for added base. Conductivity is achieved with hydroxide and sodium ions produced from hydrolyzed sodium formate, while sodium hydroxide byproduct is produced at the cathode.
The Na-DFFC produces a 33 mW cm–2 peak power density at 60 ºC and operates continuously for 13 hours at a constant 10 mA current. Contrary to the conventional chlor-alkali process, the Na-DFFC generates electricity and base simultaneously.
The team suggests that the formate fuel used to power the fuel cell may be synthesized photocatalytically from atmospheric carbon dioxide; thereby producing electricity from existing chemical feedstocks without environmental pollution.
- A Sodium-Ion-Conducting Direct Formate Fuel Cell: Generating Electricity and Producing Base,
Yinshi Li, Ying Feng, Xianda Sun, Yaling He,
Angew. Chem. Int. Ed. 2017, 56, 5734–5737.
DOI: 10.1002/anie.201701816