Intramolecular rearrangement reactions are important in organic synthesis. However, they are challenging to control because the rate of such reactions is independent of the mixing speed or the concentration of the reactants. The transitory intramolecular rearrangement of chemical intermediates proceeds very quickly even at –78 oC, which is slightly above the melting point of many organic solvents.
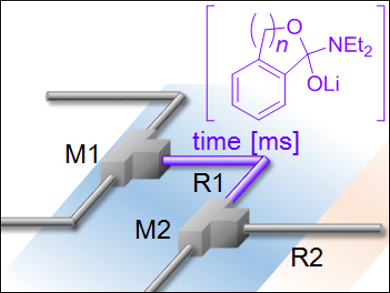
Jun-ichi Yoshida and colleagues, Kyoto University, Japan, have discovered that the anionic Fries-type rearrangement of carbamoyl-substituted aryllithium intermediates can be controlled using a flow microreactor (pictured). Precise residence times and temperatures (–25 to –50 oC) were used to trap unrearranged or rearranged intermediates, which were subsequently reacted with acyl chloride electrophiles to yield [1,4]- and [1,5]-rearranged products selectively.
Systematic experiments on [1,3]-, [1,4]-, [1,5]-, and [1,6]-anionic Fries-type rearrangements revealed that the rearrangement rate increases in the order [1,3]<[1,6]≪[1,4]≤[1,5]. Cross-over products, which would be indicative of intermolecular carbamoyl group migration, were not detected. The team suggests flow microreactors could allow transformations beyond the reach of batch reactors.
- Harnessing [1,4], [1,5], and [1,6] Anionic Fries-type Rearrangements by Reaction-Time Control in Flow,
Heejin Kim, Keita Inoue, Jun-ichi Yoshida,
Angew. Chem. Int. Ed. 2017.
DOI: 10.1002/anie.201704006