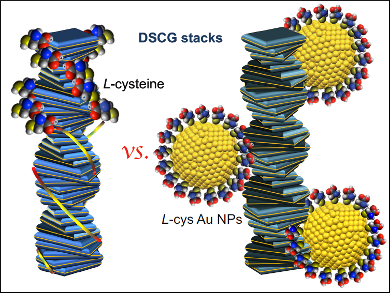
Chirality is a key scientific concept present everywhere in nature. Chirality emanating from surfaces plays an important role in studies trying to explain the origin of homochirality on our planet. Interactions between chiral molecules and surfaces (some at the nanoscale) have been shown to drive the formation of homochirality.
Leah Bergquist and Torsten Hegmann, Liquid Crystal Institute, Kent State University, OH, USA, investigate the transfer of chirality from an adsorbed molecule to a metal nanoparticle surface, and from the resulting chiral nanoparticle to its surroundings. The researchers compared the interactions of either the chiral amino acid L-cysteine or gold nanoparticles capped with chiral ligands with lyotropic chromonic liquid crystals (LCLCs, pictured). The LCLCs are formed by the asthma drug Cromolyn (disodium cromoglycate) in water. The assembly of these flat ionic molecules into stacks resembles the self-assembly of DNA (which is also an LCLC), with an intra-columnar twist as well as a twist among stacks as manifestations of induced chirality.
The results show that the intrinsic chirality of nanoscale particles is able to generate a stronger chiral response in the medium surrounding them (a chiral twist within and among stacks) than the free chiral amino acid, while the number of chiral ligands is several orders of magnitude lower.
- Chiral Amplification by L-Cysteine-Capped Gold Nanoparticles in Lyotropic Chromonic Liquid Crystals,
Leah Bergquist, Torsten Hegmann,
ChemNanoMat 2017.
DOI: 10.1002/cnma.201700286