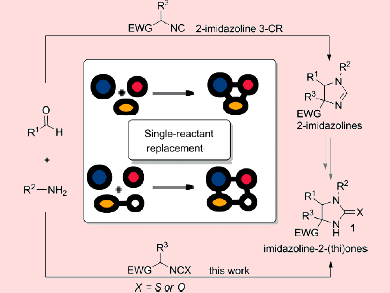
It is very difficult to predict the reactivity in multicomponent reactions (MCRs). The reactions proceed by multiple pathways and are associated with a large variety of reactants and functional groups. Theoretical chemistry has been used to verify experimental observations but not in the early stages of the development of a (multicomponent) reaction process.
F. Matthias Bickelhaupt and Romano V. A. Orru, Vrije Universiteit Amsterdam, The Netherlands, and colleagues think that a pragmatic approach of iterative theory/experiment cycles can change this. Therefore, the team has truly integrated theoretical and synthetic chemistry. By doing so, they identified in full detail the complex and complicated reaction characteristics of the novel MCR of α-acidic iso(thio)cyanates, amines, and aldehydes to access densely functionalized imidazoline-2-(thi)ones.
The team observed a lack of reactivity for isocyanates. They used a model system to understand the reaction profile and found out that the difference in proton affinity between the imine and the iso(thio)cyanate is the limiting parameter of the reaction. This finding was then used to computationally determine the scope and limitations and, thus, the performance of the reaction for a larger combinatorial set of possible inputs. Differences between the theoretical and experimental trends led to refinement of the reaction profile, that is C–C bond formation as a second decisive parameter.
According to the researchers, the predictive ability of the approach is a great advantage to the development of new reactions.
- Integrative Theory/Experiment-Driven Exploration of a Multicomponent Reaction towards Imidazoline-2-(thi)ones,
Art Kruithof, Jos R. Mulder, J. M. Ruiz, Elwin Janssen, M. Mooijman, Eelco Ruijter, Celia Fonseca Guerra, F. Matthias Bickelhaupt, Romano V. A. Orru,
Eur. J. Org. Chem. 2017.
https://doi.org/10.1002/ejoc.201700941