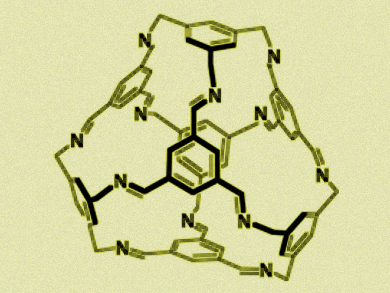
Increasing interest has been paid to shape-persistent organic cages. Cages with different shapes and functions can be obtained by applying dynamic covalent bond formations, such as imine or boronic ester condensation. Shape-persistent organic cages can be used as soluble units for porous materials. Due to their intrinsic solubility, porosity can be processed into thin films, membranes or even realized in the liquid aggregation state. However, the influence of the precursor’s rigidity on cage formation is still not well understood.
Michael Mastalerz and colleagues, Ruprecht-Karls-Universität Heidelberg, Germany, have created truncated tetrahedral [4+4] imine cages (pictured) via the condensation of aromatic trialdehydes and triamines. Both reactants were equipped either with ethyl groups to suppress the rotation of functional groups and keep them directed for the imine formation or with hydrogen atoms to allow free rotation of the reacting groups.
It was found that, in at least one of the precursors, the rotation of reacting groups must be substantially hindered to form the cages. Otherwise, an insoluble polymeric material is formed. Besides designing a new type of cage geometry, fundamental insight into the need for precursor rigidity was gained. According to the researchers, this will be helpful to construct new organic cages with other geometries, sizes, and shapes.
- Shape-Persistent [4+4] Imine Cages with a Truncated Tetrahedral Geometry,
Jochen C. Lauer, Wen-Shan Zhang, Frank Rominger, Rasmus R. Schröder, Michael Mastalerz,
Chem. Eur. J. 2018.
https://doi.org/10.1002/chem.201705713