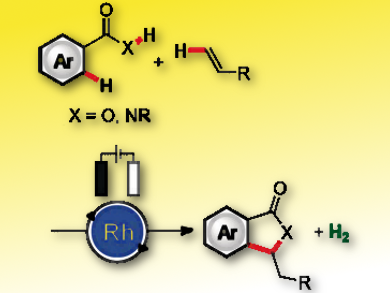
Rhodium(III) catalysts play an historic role in oxidative C–H functionalization chemistry. It typically requires stoichiometric amounts of toxic and/or expensive sacrificial metal oxidants such as silver(I) and copper(II) salts. Electrosynthesis uses electricity as the sole oxidant, thereby removing the need for chemical oxidants.
Lutz Ackermann and colleagues, Georg-August-Universität Göttingen, Germany, have discovered the first electrochemical rhodium-catalyzed C–H activation that avoids use of hazardous chemical oxidants. The environmentally benign method uses electricity as a terminal oxidant to perform cross-dehydrogenative twofold C–H/C–H functionalization with weakly coordinating benzoic acids and benzamides, generating H2 as the sole byproduct. The method tolerates a range of electron-deficient arenes and alkenes, including sensitive esters and enolizable ketones.
Evidence suggests that the Rh(III)-catalyzed electrooxidative C–H alkenylation occurs by a base-assisted internal electrophilic substitution (BIES) mechanism, within which a potassium acetate additive has a significant influence on the reduction potential of a key organometallic Rh(III) species.
- Electrooxidative Rhodium-Catalyzed C–H/C–H Activation: Electricity as Oxidant for Cross-Dehydrogenative Alkenylation,
Youai Qiu, Wei-Jun Kong, Julia Struwe, Nicolas Sauermann, Torben Rogge, Alexej Scheremetjew, Lutz Ackermann,
Angew. Chem. Int. Ed. 2018.
https://doi.org/10.1002/anie.201803342