Pt‐based materials are effective catalysts for the oxygen reduction reaction (ORR). The mechanism is complex even on a clean, unstrained Pt(111) surface and the experimental observations of reactivity on real, i.e., core–shell nanoparticles, are even more complicated because of the phase segregation in the nanoparticle. Understanding the behaviour of oxygen (O) and hydroxyl (OH) on strained Pt(111) is important to establish a framework around which the ORR mechanism on strained nanoparticle surfaces can be discussed.
Ian G. Shuttleworth, Nottingham Trent University, UK, has investigated how one group of modern catalysts (PtxM1–x) responds at a microscopic level during the oxygen reduction reaction (ORR). Shuttleworth used density functional theory (DFT) calculations to look at how the oxygen and hydroxyl species bind to the Pt(111) surfaces of these catalysts under mechanical strain, i.e., compression.
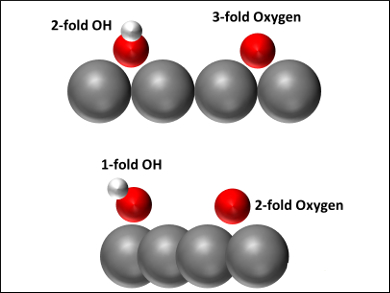
The simulations showed that the oxygen atoms and hydroxyl groups change their surface valence when the mechanical state of the surface Pt atoms changes during compression (pictured, compressed state bottom). The oxygen binding position changes from an FCC (i.e., three-fold bridge) site to a two-fold bridge site, while the hydroxyl binding position of the OH changes from a two-fold bridge site to an on‐top site. This change in binding position causes changes to the magnetization of the O‐ and OH‐populated surfaces, which results in electronic changes. Overall, the work shows that typical strains cause significant changes on the surfaces of nanoparticles with important applications.
- Binding Site Transitions Across Strained Oxygenated and Hydroxylated Pt(111),
Ian G. Shuttleworth,
ChemistryOpen 2018.
https://doi.org/10.1002/open.201800039