Drug resistance in pathogens such as the Gram-negative bacterium Pseudomonas aeruginosa becomes more and more frequent. P. aeruginosa can cause life‐threatening infections in immunocompromised patients. However, the antibiotic development pipeline is currently drying out. The virulence of these organisms is controlled by cell-to-cell communication, so the inhibition of this crosstalk holds promise for the discovery of new pharmaceuticals.
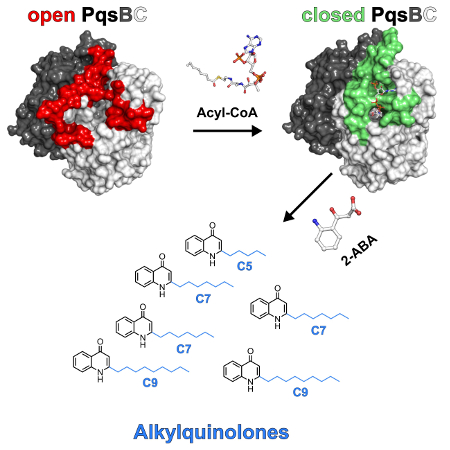
P. aeruginosa produces many saturated and mono‐unsaturated 2‐alkyl‐4(1H)‐quinolones (AQs) and AQ N‐oxides (AQNOs) that serve as signaling molecules to control the production of virulence factors. The β‐ketoacyl‐acyl‐carrier protein synthase III (FabH)‐like heterodimeric enzyme PqsBC catalyzes the last step in the biosynthesis of the most abundant AQ congener, 2‐heptyl‐4(1H)‐quinolone (HHQ), by condensing octanoyl‐coenzyme A (CoA) with 2‐aminobenzoylacetate (2‐ABA). However, the basis for the large number of other AQs/AQNOs produced by P. aeruginosa is not known.
Wulf Blankenfeldt, Helmholtz Centre for Infection Research and University of Braunschweig, Germany, and colleagues have investigated PqsBC. PqsBC uses different medium‐chain acyl‐CoAs (ubiquitous coenzymes that play a major role in fatty acid metabolism) to produce various saturated AQs/AQNOs. It also biosynthesizes mono‐unsaturated congeners.
A comparison of several crystal structures showed that PqsBC adopts open, intermediate, and closed conformations that change the shape of the acyl‐binding pocket. Such structural transitions had been postulated but had never been observed. They could provide an explanation for the catalytic cycle of PqsBC and may be used to develop specific path blockers.
- The Alkylquinolone Repertoire of Pseudomonas aeruginosa is Linked to Structural Flexibility of the FabH-like 2-Heptyl-3-hydroxy-4(1H)-quinolone (PQS) Biosynthesis Enzyme PqsBC,
Florian Witzgall, Tobias Depke, Michael Hoffmann, Martin Empting, Mark Brönstrup, Rolf Müller, Wulf Blankenfeldt,
ChemBioChem 2018.
https://doi.org/10.1002/cbic.201800153