Theoretically, steering biological processes and systems with light allows precise control without permanently contaminating the target. There is, however, one fundamental problem: most biomolecules are not sensitive to light (apart from their eventual degradation). Thus, artificial “adapters” are needed to transform light energy into biological functions. Many of these systems have the additional problem that they only respond to harmful UV light.
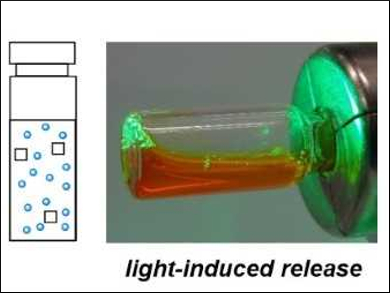
Zbigniew L. Pianowski and Johannes Karcher, Karlsruhe Institute of Technology, Germany, have created a light-sensitive biocompatible material that forms supramolecular hydrogels. The used hydrogelator is based on a photochromic (S)‐α‐amino acid, which was coupled to a protected (S)‐lysine. The resulting hydrogels can physically encapsulate various unmodified drug molecules. Upon irradiation with green (i.e., visible) light, the gels dissipate to fluid form and release the encapsulated drugs in their active form. This effect is due to a transition of the majority of the hydrogelator’s molecules from the trans‐ to the cis‐isomer, which destabilizes the hydrogel’s structure.
Using a gel loaded with antibiotics, the growth of bacterial cultures could be suppressed with green light. The developed gels could have applications in photopharmacology, for example, in the form of light-induced drug-releasing implants or, after microgel formulation, for controlled drug delivery systems delivered through the bloodstream.
- Photocontrol of Drug Release from Supramolecular Hydrogels with Green Light,
Johannes Karcher, Zbigniew L. Pianowski,
Chem. Eur. J. 2018, 24, 11605–11610.
https://doi.org/10.1002/chem.201802205