The photoelectrochemical reduction of CO2 to form fuels could help to reduce global warming. Compared to possible gaseous products, liquids such as methanol are easier to store and transport. However, methanol production involves six-proton-coupled six-electron-transfer processes, which makes this reaction kinetically sluggish. This means it is difficult to selectively produce methanol by CO2 reduction. It is especially challenging in aqueous electrolytes, where proton reduction (i.e., hydrogen production) is a highly competitive reaction.
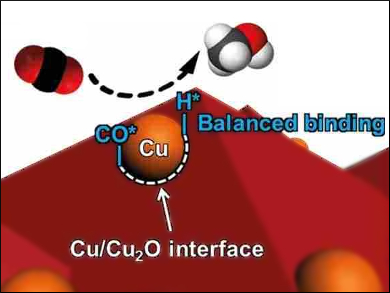
Jinlong Gong, Tianjin University, China, and colleagues have developed a cathode for the increased photoelectrochemical production of methanol. The team created a well-defined Cu/Cu2O interface by introducing metallic Cu nanoparticles onto Cu2O films via electron-beam evaporation. The Cu/Cu2O interfaces (pictured) greatly enhance the binding of surface‐adsorbed hydrogen atoms (H*) and reduce the binding of CO*. This provides balanced amounts of H* and CO* intermediates for methanol production.
In a photoelectrochemical cell with a TiO2 model photoanode, a Cu/Cu2O dark cathode has a Faradaic efficiency of up to 53.6 % for methanol production. According to the researchers, this approach provides a method for high‐efficiency, low‐cost, and stable methanol generation.
- Tuning Cu/Cu2O Interfaces for the Reduction of Carbon Dioxide to Methanol in Aqueous Solutions,
Xiaoxia Chang, Tuo Wang, Zhi-Jian Zhao, Piaoping Yang, Jeffrey Greeley, Rentao Mu, Gong Zhang, Zhongmiao Gong, Zhibin Luo, Jun Chen, Yi Cui, Geoffrey A. Ozin, Jinlong Gong,
Angew. Chem. Int. Ed. 2018.
https://doi.org/10.1002/anie.201805256