Redox transmetallation protolysis (RTP) reactions of free rare earth metals are a convenient and efficient route to highly reactive rare earth compounds. Usually, these reactions use diarylmercury reagents, particularly Hg(C6F5)2. However, these reagents are toxic and environmentally unfriendly.
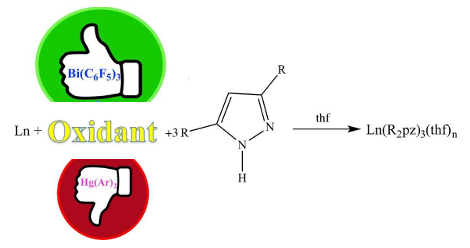
Peter C. Junk, James Cook University, Townsville, QLD, Australia, and colleagues have shown that the diarylmercury compounds in RTP reactions can be replaced by tris(pentafluorophenyl)bismuth, Bi(C6F5)3, in the synthesis of a wide range of pyrazolate complexes (pictured below, pz = pyrazolate, thf = tetrahydrofuran).
The pyrazolatolanthanoid(III) complexes were prepared by reactions between a lanthanoid metal, Bi(C6F5)3, and the respective pyrazoles. The products were characterized by 19F NMR and infrared (IR) spectroscopy, microanalyses, and X-ray crystallography.
Bi(C6F5)3 was shown to be an excellent oxidative reagent for RTP reactions with lanthanoid metals and a range of 3,5-disubstituted pyrazoles. The reaction works with a number of Ln metals and a variety of pyrazoles. According to the researchers, using Bi(C6F5)3 should make RTP reactions more attractive as a preparative method.
- Can Bismuth Replace Mercury in Redox Transmetallation/Protolysis Syntheses from Free Lanthanoid Metals?,
Zhifang Guo, Victoria Blair, Glen B. Deacon, Peter C. Junk,
Chem. Eur. J. 2018, 24, 17464–17474.
https://doi.org/10.1002/chem.201804703




