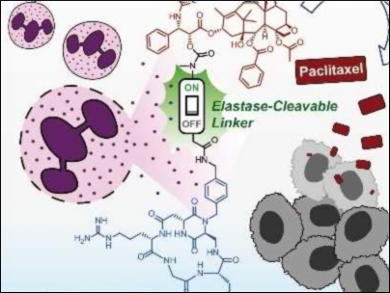
Anticancer drugs usually have significant side effects. Restricting their activity to the site of a tumor can help to prevent some of the harmful effects. One strategy for this is binding the drugs to ligands which target some component that is specific to cancer cells.
One such component is αVβ3, a transmembrane receptor which is overexpressed in a variety of cancers. The conjugation of drugs to small-molecule ligands that tightly bind to integrin αVβ3 is an important tumor-targeting strategy. However, this can reduce the potency compared with the free drug.
Cesare Gennari, Milan University, Italy, and colleagues have found an integrin ligand which accumulates on the membrane of αVβ3-expressing cancer cells. The team connected this ligand to the anticancer drug paclitaxel via an Asn‐Pro‐Val (NPV) tripeptide linker. This linker can be severed by neutrophil elastase, an enzyme secreted by a type of white blood cell. Neutrophils are a hallmark of inflammation and naturally occur around many types of tumors.
Severing the peptide linker triggers the release of paclitaxel outside the cancer cells. The free drug then enters the cells by passive diffusion with no loss of potency. According to the researchers, this type of treatment could be therapeutically active against a large variety of tumor types.
- Neutrophil Elastase Promotes Linker Cleavage and Paclitaxel Release from an Integrin-Targeted Conjugate,
André Raposo Moreira Dias, Arianna Pina, Amelia Dean, Hans-Georg Lerchen, Michele Caruso, Fabio Gasparri, Ivan Fraietta, Sonia Troiani, Daniela Arosio, Laura Belvisi, Luca Pignataro, Alberto Dal Corso, Cesare Gennari,
Chem. Eur. J. 2018.
https://doi.org/10.1002/chem.201805447