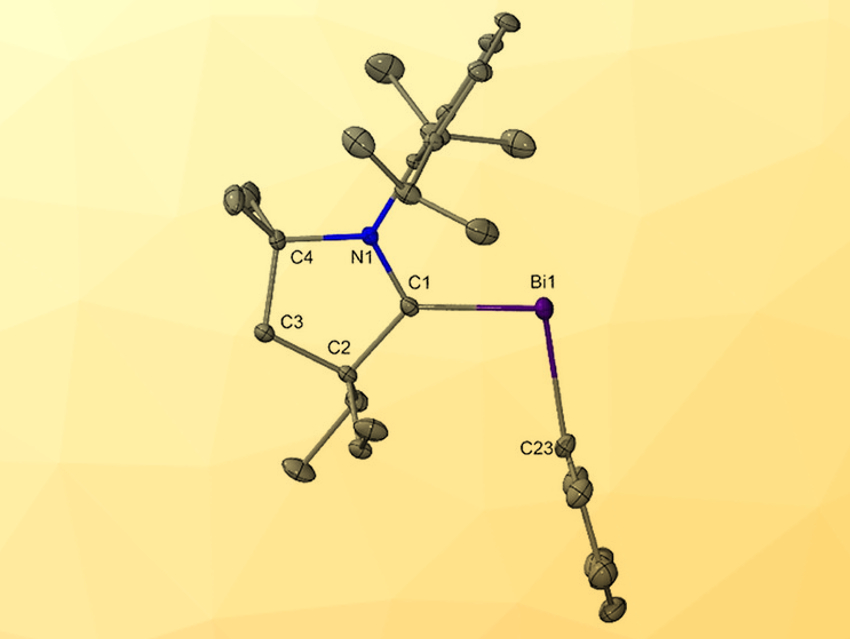
Carbene-pnictinidenes (group 15 compounds) have the general structure L–E–R (L = carbene; E = N, P, As, Sb; R = H, halide, alkyl, aryl). These compounds have a variety of applications in chemical synthesis. However, the bismuth analogs of this type of compound (carbene-bismuthinidenes) had not yet been synthesized.
Zoltán Benkő, Budapest University of Technology and Economics, Hungary, Robert J. Gilliard, Jr., University of Virginia, Charlottesville, USA, have synthesized and structurally characterized this elusive carbene-bismuthinidene (pictured above). The compound was prepared from bismuth precursors via Route A or B (pictured below), using a beryllium(0) complex as a reducing agent and/or ligand transfer reagent.
This is the first example of a carbene-bismuthindene and features the shortest Ccarbene–Bi bond to date. The first use of molecular Be(0) to prepare subvalent organometallic complexes is also significant. These findings may lead to new applications for both bismuth and beryllium in other areas of organometallic chemistry, such as bond activation and catalysis.
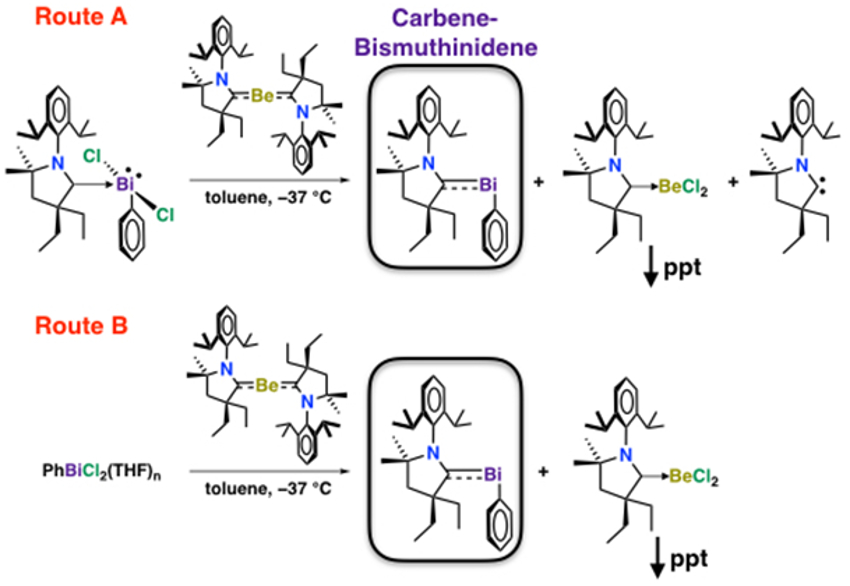
- Isolation of Cyclic(Alkyl)(Amino) Carbene-Bismuthinidene Mediated by a Beryllium(0) Complex,
Guocang Wang, Lucas A. Freeman, Diane A. Dickie, Réka Mokrai, Zoltán Benkő, Robert J. Gilliard Jr.,
Chem. Eur. J. 2019.
https://doi.org/10.1002/chem.201900458