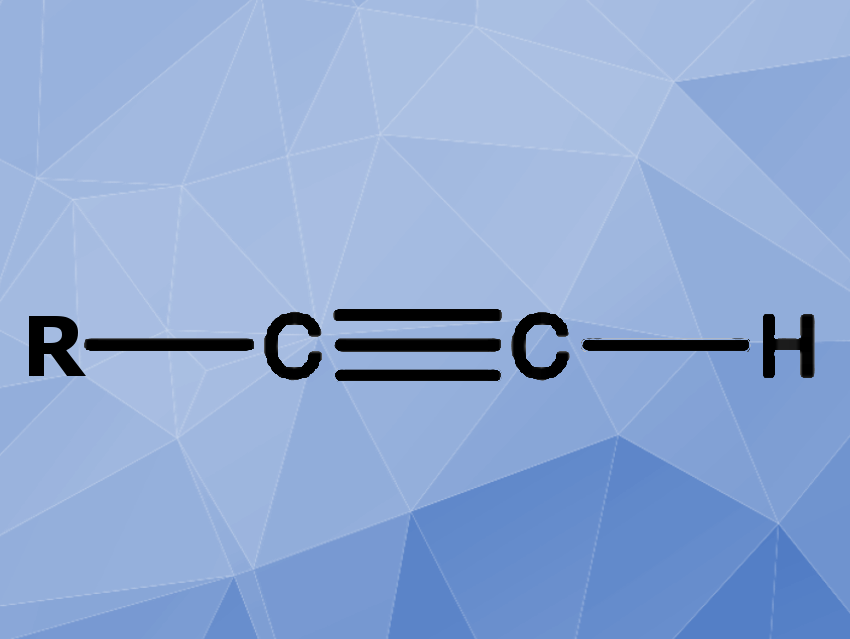
A research team from the University of California, Berkeley, CA, USA, has discovered and characterized the genes and proteins used by a soil bacterium to produce terminal-alkyne-containing amino acids. Terminal alkynes are used for click-chemistry syntheses of many drug-related components, and the discovery may open new strategies for easier modifications on peptides or proteins used in medicine and technology.
Click Chemistry
The term “click chemistry”, which was coined by Barry Sharpless, is the rapid reaction of a terminal alkyne with an azide under mild conditions to form a triazole. The triazole then offers a set of follow-up reactions so that the original alkyne group might end up as a sophisticated two-carbon entity in the middle of a complex organic molecule, which can be a drug, a natural product, or a useful catalyst ligand.
Click-chemical approaches are very popular in organic synthesis, but they were not applicable for natural peptides and proteins, simply because biochemical pathways leading to terminal-alkyne-containing amino acid were not known. Of course, it would be possible to synthesize such amino acids manually and blend them into a peptide through automated peptide synthesis, but the methods are elaborate and unsuitable for larger proteins.
Two Natural Amino Acids with an Alkyne Group
However, natural amino acids containing an alkyne group are not entirely unknown: Screenings for antibiotically active substances carried out in the 1970s and 1980s have revealed that some fungi and bacteria produce an alkyne-carrying serine, β-ethynylserine, or an alkynyl glycine, L-propargylglycine. But how do these microbiotic organisms synthesize the alkynyl-modified amino acids? If they knew the biological pathway, researchers could start to explore biotechnological strategies to create the amino acids in model organisms and build peptides from them.
Michelle Chang and her colleagues focused on the bacterium Streptomyces cattleya to examine its synthesis of β-ethynylserine. Employing elaborate biochemical and microbiological approaches, including the identification and characterization of each of the participating bacterial enzymes, the researchers eventually identified all necessary steps, genes, and enzymes.
It all Begins with Lysine
The pathway starts with the natural amino acid L-lysine, which is oxidized and chlorinated at its β-carbon by a halogenase, then ammonia and formaldehyde are cleaved off to form a 4-chloro-allylglycine, which is then dechlorinated to yield L-propargylglycine, the first terminal-alkyne amino acid synthesized on that pathway.
Hydroxylation in α-position to the amino group would then yield β-ethynylserine. However, this process proved to be less straightforward then the other biosynthetic steps. The researchers reported that the enzyme thought to catalyze that step did not recognize L-propargylglycine as a substrate. Hydroxylation only worked out when the glycine was coupled to glutamate to form a dipeptide, which apparently was the substrate. With all these steps characterized, the researchers had for the first time identified a biosynthetic pathway yielding natural terminal-alkyne amino acids.
Now, Make Peptides
With the biochemical pathway identified and the involved proteins and their genes characterized, it would be possible, in principle, to turn to biotechnology and reconstruct the pathway in a model organism. Therefore, the researchers transformed Escherichia coli and, indeed, made it produce proteins containing the modified amino acid, as they confirmed by proteome analysis.
Further applications could involve bioorthogonal synthetic strategies: Endogeneously synthesized peptides could be engineered containing terminal-alkyne amino acids, which could be further used for click reactions, metathesis reactions, polymerization, and other bioorthogonal chemistries.
- Discovery of a pathway for terminal-alkyne amino acid biosynthesis,
J. A. Marchand, M. E. Neugebauer, M. C. Ing, C.-I. Lin, J. G. Pelton, M. C. Y. Chang,
Nature 2019, 567, 420–424.
https://doi.org/10.1038/s41586-019-1020-y