The malaria parasite invades, digests, and reproduces in the host’s red blood cells. During this process, the parasite degrades the peptides in hemoglobin, oxidizes the heme group’s central atom to iron(III), and crystallizes the resulting FeIII(protoporphyrinato) compound to give the excretion product “malaria pigment”. This process is inhibited by many existing antimalarial drugs.
Scott Bohle, McGill University, Montreal, Canada, Peter Stephens, State University of New York, USA, and colleagues, have synthesized a new crystallized synthetic heme derivative, deuterohematin anhydride (DHA), which differs from the malaria pigment only by the absence of vinyl groups on the porphyrin ring. DHA was prepared by the dehydrohalogenation of deuterohemin with the non-coordinating base 2,6-lutidine.
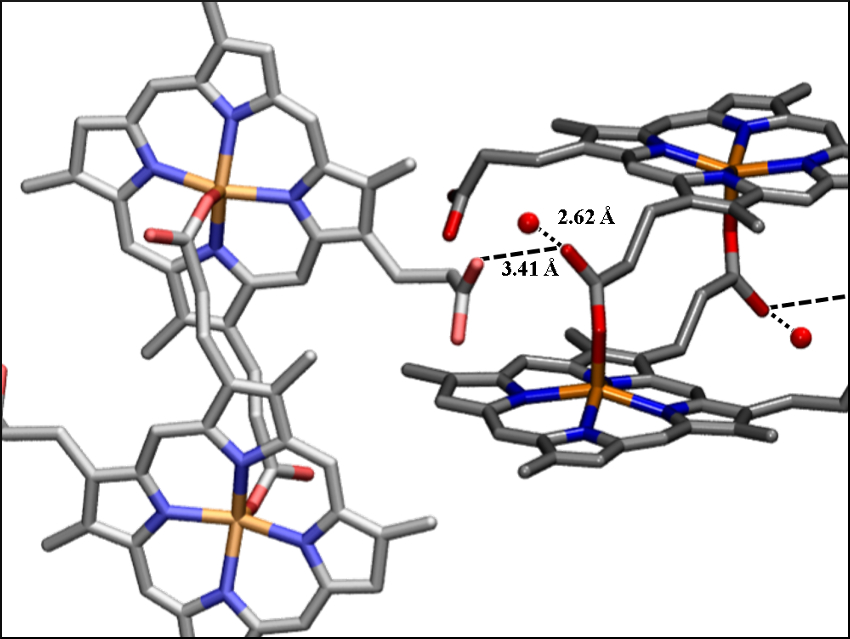
The team used X-ray powder diffraction to determine the structure of DHA. Like malaria pigment, it consists of propionate-bound heme dimers. However, the dimers do not form a chain as in the natural product. In contrast to malaria pigment, the propionate groups in DHA are twisted or bent and two water molecules fit into the gap between the propionates (pictured). Because of these structural differences, DHA has an increased solubility compared with malaria pigment.
The researchers have identified an infrared spectroscopic (IR) signature band for this structural motif and found that many preparations of malaria pigment have this IR band in their amorphous coproducts. These results suggest that this is a promising new target for the next generation of antimalarial drugs: If heme crystallization was directed towards DHA-like phases and away from the malaria pigment structure, then the increased solubility of the heme could disrupt the growth of the malaria parasite.
- Hydrating the Bispropionate Notch in Malaria Pigment: A New Structural Motif in the Iron(III)(deuteroporphyrin) Dimer,
David Kuter, Liliana Suárez, Erin L. Dodd, Bruce C. Noll, Peter W. Stephens, D. Scott Bohle,
Chem. Eur. J. 2019.
https://doi.org/10.1002/chem.201805116