150 years ago Dmitri Ivanovich Mendeleev formulated the periodic system, a fundamental law of chemistry which eventually was explained in terms of quantum physics. In the spring of 1869, 34-year-old D. I. Mendeleev presented his first version of what subsequently became known as the periodic system or table to the newly established Russian Chemical Society. I shall use the two terms periodic system or table synonymously.
1. Macroscopic Representation of the Atom’s Internal Structure
Mendeleev’s system or classification of the chemical elements provided inorganic chemistry with a solid foundation which brought order to the confusing amount of experimental data; at the same time, it predicted the existence of several hitherto unknown elements. In short, it proved immensely fertile.
Although the present versions of the periodic table contain 118 elements compared to the 63 in Mendeleev’s original table, the systems used today descend directly from the one proposed in 1869. Until the end of the nineteenth century, the periodic system belonged exclusively to the science of chemistry, but when it came to explaining why the system works so well, it were physicists who took the lead. As it turned out in the 1920s, the periodic system is a macroscopic representation of the internal structure of the atoms. From this point of view, it is as much about physics as it is about chemistry.
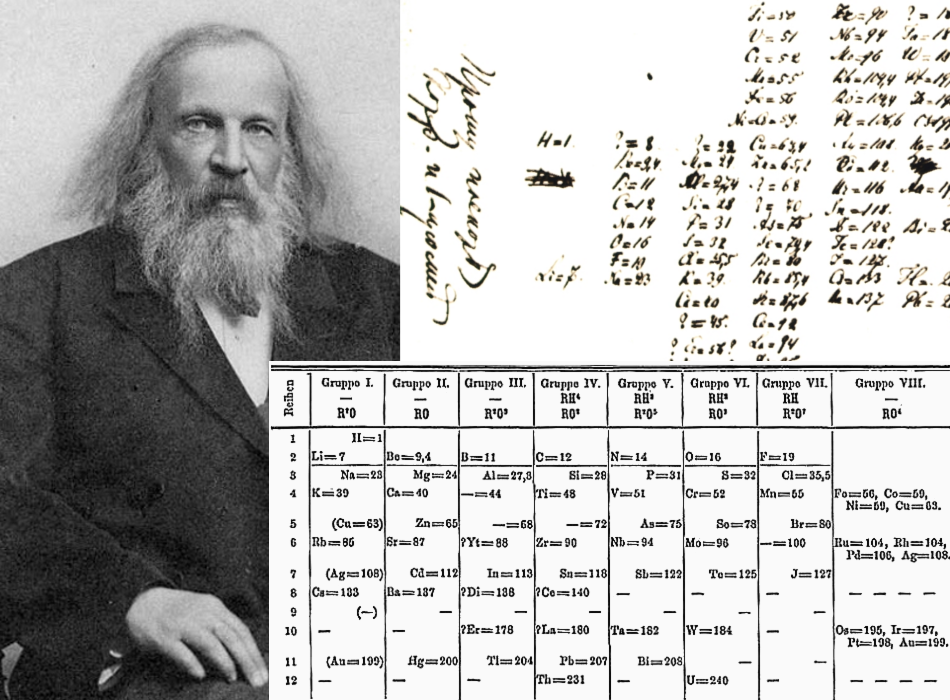
Figure 1. Dmitri Ivanovich Mendeleev (1834 – 1907) and his periodic table.
2. Chemical Elements
Of course, the periodic system presupposes knowledge of what constitutes a chemical element. The modern concept of an element goes back to the late eighteenth century and owes much to the French chemist Antoine-Laurent Lavoisier, the great reformer of chemistry. Lavoisier’s definition of an element was empirical in so far that he limited elements to substances that could not be decomposed to still simpler substances. While this definition is independent of whether matter consists of atoms or not, in 1808 John Dalton took the important step to link together elements and atoms.
According to Dalton, an atom is the smallest unit of an element, meaning that there are as many different atoms as there are different elements. Moreover, Dalton introduced the crucial idea of atomic weight, thereby associating an element with a measurable quantity. A pure element could thus be defined as a substance with a particular atomic weight.
By the late 1850s, when Dalton’s ideas had become widely accepted, determinations of atomic weights were an important part of chemistry. It was from this basis that Mendeleev constructed his system of the elements, and it was from the same basis that the German chemist Julius Lothar Meyer independently suggested a roughly similar scheme later in 1869. The periodic system thus has two fathers, but Mendeleev came first and his system was more influential than Meyer’s.

Figure 2. Julius Lothar Meyer (1830 – 1895) and Laurent Lavoisier (1743 – 1794).
3. Predictions, Successes, and Problems
3.1. Advantages of Mendeleev’s Periodic Table
By the mid-1880s, the periodic system was recognized to be an indispensable key to understand the elements and their combinations into chemical compounds. There were basically two major reasons why chemists responded so positively to Mendeleev’s innovation: First, it arranged all the known elements in one coherent system and, thus, established for the first time an organizing principle valid for all the elements and their properties.
Second and even more importantly, Mendeleev’s system had a remarkable predictive power. Not only did it predict the existence of new elements, which were subsequently discovered, but it also revised the atomic weight of some of the elements. For example, the atomic weight of beryllium was thought to be approximately 14.5, which would place the element as a homologue of aluminium, whereas Mendeleev argued that his system required the atomic weight to be close to 9. When experiments from about 1880 proved beryllium’s atomic weight to be 9.1, naturally it was considered a triumph for Mendeleev’s classification system.
As regards the prediction of new elements, Mendeleev famously claimed that gaps in his system corresponded to three unknown elements with atomic numbers ca. 44, 68, and 72 and resembling boron, aluminium, and silicon, respectively. When the three elements were discovered in nature between 1875 and 1886, they were named scandium (1879), gallium (1875), and germanium (1886), and their properties proved to correspond closely to those predicted by Mendeleev.
To most chemists, these and other successful predictions proved the correctness of the periodic system beyond any reasonable doubt. However, several of Mendeleev’s predictions were quite wrong, such as his prediction of “eka-caesium” of atomic weight ca. 175 and his later suggestion that the physicists’ ether was a chemical element much lighter than hydrogen.
2.2. Noble Gases
Despite its many successes, by the late nineteenth century, the periodic system faced problems and anomalies indicating flaws in the original system. For example, it followed from Mendeleev’s table that iodine must necessarily have an atomic weight higher than tellurium, and yet experiments showed the opposite. When argon was unexpectedly discovered in 1894 and turned out to be a monatomic gas with atomic weight 39.8, it created problems. There seemed to be no place for the new element in the periodic system, which at first caused Mendeleev to doubt its existence and suggest that perhaps it was not an element but rather triatomic nitrogen N3. With the discovery of helium and other noble gases, it was realized that the threat to the periodic system only seemed to exist. All that was needed was to extend the original system with a new group reserved for the noble gases.
Nonetheless, it remained a problem that the atomic weight of argon was between those of potassium (39.1) and calcium (40.1). Might it be that the atomic weight was not, after all, the proper ordering parameter for the periodic system?
4. Early Attempts at Explanation
4.1. Atomic Structure and Weights
According to Mendeleev and most chemists, the periodic system of the elements was first and foremost an empirical law not in need of further explanation in terms of, for example, the structure of atoms. In fact, Mendeleev emphatically denied that atoms could be composed of smaller particles. Yet, a few speculatively minded chemists wondered why the atomic weights represented in the system had certain values while other values were missing. For example, why do the elements in the first period have atomic weights approximately 7 (Li), 9 (Be), 11 (B), 12 (C), 16 (O), and 19 (F), while there are none with weights close to 8, 10, 13, 15, and so forth?
While Mendeleev dismissed such questions as nonsense, other chemists speculated that all atoms were conglomerates of very small primordial particles, either hydrogen atoms or something smaller. To this group belonged William Crookes in London, UK, Julius J. Thomsen in Copenhagen, Denmark, and also Lothar Meyer in Tübingen, Germany, who thought that the periodic system might be explained in terms of atomic structure. However, their vague and qualitative suggestions were not welcomed by the majority of chemists. It was only with J. J. Thomson’s discovery of the electron at the end of the century that they were transformed into a quantitative theory and then by a physicist rather than a chemist.
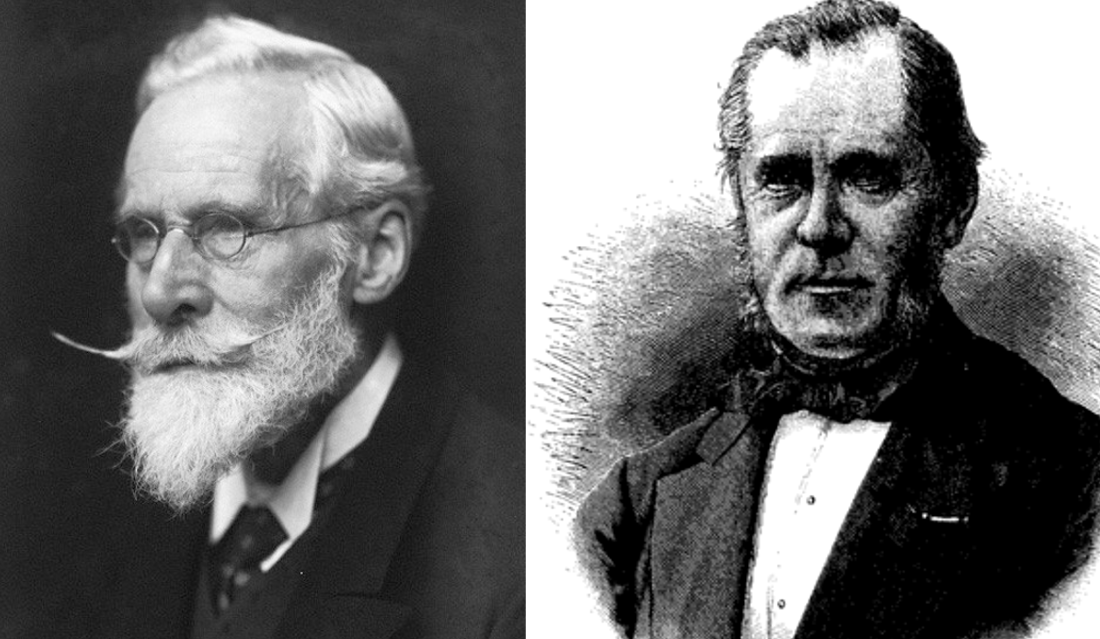
Figuer 3. William Crookes (1832 – 1919) and Julius J. Thomsen (1826 – 1909).
4.2. Thomson’s Atomic Model
According to Thomson’s atomic model from about 1904, the atoms consisted of a large number of negatively charged electrons moving in circular orbits inside a positively charged weightless sphere. On the basis of this picture, Thomsen performed complex calculations to find the stable electron configurations which he supposed to be analogous to those of real atoms. In this way, he arrived at configurations which showed a conspicuous periodicity of the same kind as Mendeleev’s table. Or, as the chemist Ida Freund optimistically wrote in 1904, Thomson’s theory provided “an explanation for the empirical relations between atomic weight and atomic properties embodied in the periodic law.”
According to Thomson’s model, the chemical properties of the elements were associated with certain structures of electrons which corresponded to and explained the chemical groups. However, he associated the groups with internal electron structures and not, as in later theories, with the electrons in the outermost ring. Thomson’s spirited attempt to explain the periodic system in terms of electron configurations was not only sketchy, it also built on a wrong atomic model which survived for less than a decade. Nonetheless, it was the first such attempt and, for this reason alone, deserves a place in the history of science.
5. The Nuclear Atom and Its Consequences
5.1. Radioactivity
The discovery of radioactivity in 1896 and the recognition that radioactive decay implied the instability of certain elements caused problems for the conventional understanding of elements and their classification. Mendeleev flatly denied that radioactivity was an atomic property and also that radium and other heavy elements spontaneously transmuted into other elements. This, he believed, was contrary to the very nature of elements on which the periodic law rested.
A related problem was the confusing number of radioactive substances isolated from the decay series of uranium, thorium, and actinium. Some of these substances were chemically inseparable from other elements and yet they were different from them and without their own positions in the periodic table.
5.2. Isotopes
In 1910, Frederic Soddy suggested that there were different species of the same element, with the species differing in their atomic weights. The suggestion was radical as it violated the established chemical principle that every portion of an element has the same atomic weight. As Soddy admitted, it was “in direct opposition to the principle of the periodic law.” Three years later he coined the word “isotope”, which he now related to Ernest Rutherford’s hypothesis of an atomic nucleus.
By about 1915, many physicists had converted to a new picture of the atom designed by Rutherford and Niels Bohr, including that the atomic number as given by the nuclear charge was the defining parameter of an element. Since two isotopes have the same atomic number, they occupy the same position in the periodic table despite having different atomic weights; and because the electron configuration depends only on the nuclear charge, they have the same chemical properties.
The concept of the atomic number received convincing confirmation from X-ray spectroscopic measurements pioneered by Henry Moseley. This also provided strong support of the Rutherford-Bohr model of the atom. Moseley’s method was singularly important as it led to a new interpretation of the nature of Mendeleev’s system.
6. Bohr’s Theory of the Periodic System
As early as in his atomic theory of 1913, Bohr suggested electron configurations for the light elements that roughly corresponded to the periodic system, but it was only in his later and more sophisticated theory that he developed a full-scale explanation based on the principles of the old quantum theory. In works between 1921 and 1923, he characterized the orbits of the electrons by their principal and azimuthal quantum numbers, at the time symbolized as n and k. Guided by the correspondence principle, data from X-ray spectroscopy, and the chemical properties of the elements, he assigned, for the first time, electron configurations to all the elements from hydrogen to uranium. In this way, he was able to reconstruct the periodic system including the poorly understood group of rare earth metals, which according to Bohr consisted of exactly 14 elements.
In his Nobel Prize lecture of 1922, Bohr went even further by proposing an electron configuration of the hypothetical element of atomic number 118. This element (oganesson) has recently been synthesized in nuclear reactions and, remarkably, its electron structure is believed to be the same as Bohr predicted nearly a century ago. The possible existence of transuranic elements was also discussed by the German physicist Richard Swinne, who, in 1925, assigned electron structures to elements with atomic numbers between 92 and 105.
Bohr’s reconstruction of the periodic system attracted much positive interest, if more among atomic physicists than among chemists. In the controversial case of the unknown element 72, Bohr deduced that it had to be chemically analogous to zirconium and not, as most chemists believed, a rare earth. Inspired by Bohr’s prediction, two scientists at his institute in Copenhagen, George von Hevesy and Dirk Coster succeeded in detecting the characteristic X-ray lines of the element in zirconium minerals. The discovery of hafnium, as the new element was named, was widely seen as a triumph of Bohr’s theory. But it soon turned out that the impressive theory was only a provisional answer to the riddle of explaining the periodic system.
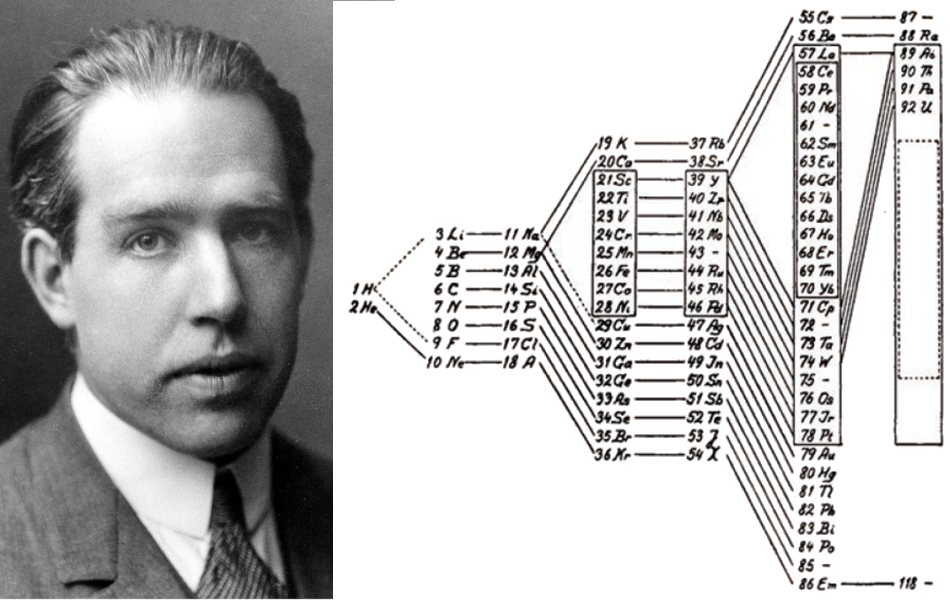
Figure 4. Niels Bohr (1885 – 1962) and his periodic table from 1922. The position of element 72 (hafnium) is still free, the hypothetical element 118 is represented as part of the group of noble gases, the symbol for element 86 (Em, emanation) refers to an older name for radon (Rn).
7. Pauli’s Exclusion Principle
Although the modern interpretation of the periodic system is based on the theory of quantum mechanics principally credited to Werner Heisenberg and Erwin Schrödinger, it goes back to a slightly earlier period. In a critical revision of Bohr’s theory, the British physicist Edmund Stoner classified in 1924 the energy levels in an atom by three quantum numbers rather than two. The result was a system of electron configurations more complete and fine-grained than Bohr’s.
Stoner’s theory stimulated Wolfgang Pauli to write a now classic paper that appeared in Zeitschrift für Physik in March 1925, in which he introduced the famous exclusion principle [1]. When Pauli belatedly was awarded the Nobel Prize in 1945, it was for his discovery of “a new law of nature, the exclusion principle or Pauli principle.”
As understood today, in its most general form the exclusion principle states that two identical fermions cannot occupy the same quantum state. Applied to atomic electrons, it states that it is impossible for two electrons in an atom to have the same values of the four quantum numbers.
One of the quantum numbers is the spin quantum number which can attain only two values corresponding to “spin up” and “spin down.” However, in March 1925 the spin had not yet been discovered nor had quantum mechanics. Pauli instead introduced what he called an electron’s “twofoldness” (Zweideutigkeit), which can be considered a precursor of the spin.
Not only did Pauli explain the details of the periodic system on this basis, but he also found that the maximum number N of elements in a period is given by N = 2n2, where n denotes the principal quantum number n = 1, 2, … The rule is known as Rydberg’s rule in honor of the Swedish physicist Janne Rydberg. In 1906, he noticed that the number of elements in the periods (2, 8, 18, and 32) can be written in this form. A similar rule was first stated by Julius Thomsen as early as 1895, but at a time when the group of noble gases was still unknown.
8. Conclusion
With the exclusion principle and the full machinery of quantum mechanics, Mendeleev’s periodic system was explained in terms of fundamental physics, at least by and large. Mendeleev, the founder of the system, would not have welcomed the development, for to him the periodic system was an independent and irreducible law of chemistry.
The article has been published in German:
- Elemente mit System,
Helge Kragh,
Physik Journal 2019, 3, 53–57.
References
[1] W. Pauli, Über den Einfluß der Geschwindigkeitsabhängigkeit der Elektronenmasse auf den Zeemaneffekt, Zeitschrift für Physik 1925, 31(1925), 373–385. http://dx.doi.org/10.1007/BF02980592
Further Reading
- M. Kaji, H. Kragh, G. Palo, Early Responses to the Periodic System, Oxford University Press, Oxford, UK 2015.
- H. Kragh, Niels Bohr and the Quantum Atom, Oxford University Press, Oxford, UK 2012.
- E. Scerri, The Periodic Table, Oxford University Press, Oxford, UK 2007.
- J. W. van Spronsen, The Periodic System of Chemical Elements, Elsevier, Amsterdam, The Netherlands 1969.




