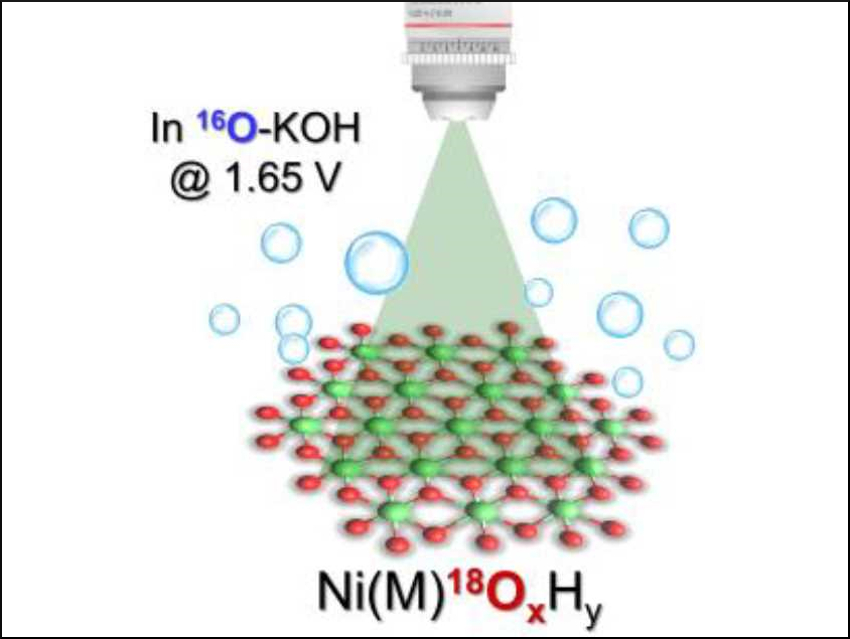
The oxygen evolution reaction (OER) is a key process in water splitting, which can be used to generate “green” hydrogen fuel. Nickel iron oxide is a benchmark catalyst for the OER in an alkaline medium. However, this catalyst is still poorly understood and the nature of its active site is debated.
Xile Hu and colleagues, Ecole Polytechnique Fédérale de Lausanne (EPFL), Switzerland, have used in-situ Raman spectroscopy and oxygen isotope experiments to study the active sites in different Ni-based oxides during electrocatalytic water oxidation. Ultrathin layered double hydroxides (LDHs) consisting of either Ni, NiCo, or NiFe oxide were synthesized and used as defined models for the catalysts. They were prepared with different oxygen isotopes: the lattice oxygen atoms were either 16O or 18O. Raman spectroscopy was then used to monitor the evolution of Raman peaks corresponding to either lattice oxygen or a Ni superoxide (NiOO–) intermediate.
The researchers found that for the pure Ni and the NiCo LDHs, lattice oxygen and NiOO– are exchanged with oxygen from OH– in the electrolyte solution during the OER. This means the lattice oxygen atoms participate in the reactions. These findings suggest that for iron-free Ni oxides, Ni sites in the bulk are the active sites and evolve oxygen via a NiOO– precursor.
In contrast, no oxygen exchange takes place for NiFe and NiCoFe LDHs. When Fe is present, the catalytic activity is dramatically increased. The researchers attribute this rise in activity to highly reactive active sites on the catalyst surface, most likely based on Fe.
- Oxygen Isotope Labeling Experiments Reveal Different Reaction Sites for the Oxygen Evolution Reaction on Nickel and Nickel Iron Oxides,
Seunghwa Lee, Karla Banjac, Magalí Lingenfelder, Xile Hu,
Angew. Chem. Int. Ed. 2019.
https://doi.org/10.1002/anie.201903200