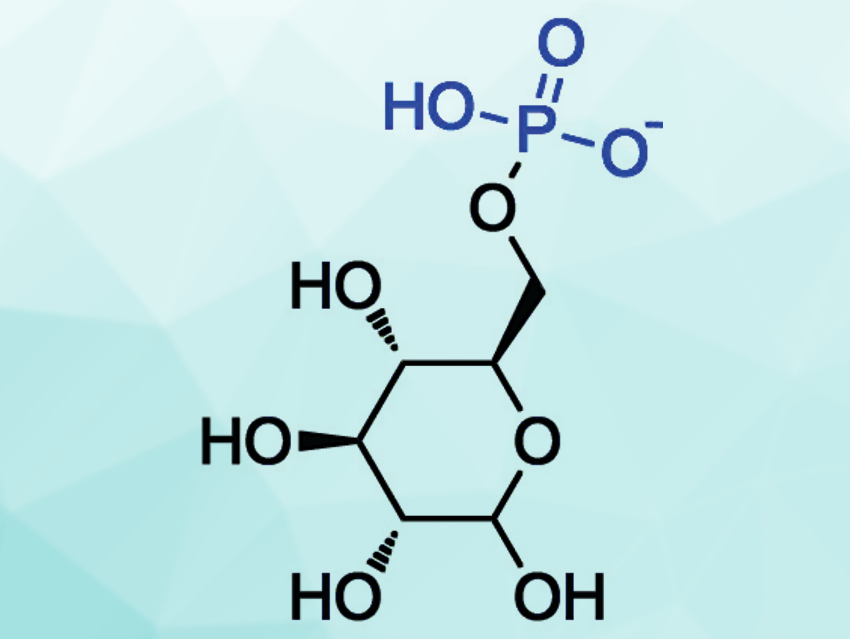
The addition of inorganic phosphates to biological substrates is important for many biochemical activities. This type of reaction is thought to proceed through a pentacoordinated phosphorane intermediate (pictured in red). Usually, the reaction requires prior activation of the phosphates. Pentacoordinated phosphoranes rapidly undergo hydrolysis in acidic media and are only detectable in basic media. This difficulty in the detection and isolation of phosphoranes has hampered their characterization.
Barnabas Otoo, Oakland City University, IN, USA, and colleagues have synthesized and characterized spontaneously formed phosphoranes of monosaccharides and polyols for the first time. These phosphoranes are prepared by the reaction of alcohols with carbonyl-activated phosphates (pictured below, first step) in the absence of activating enzymes. The phosphoranes are stabilized by cyclic acetal formation.
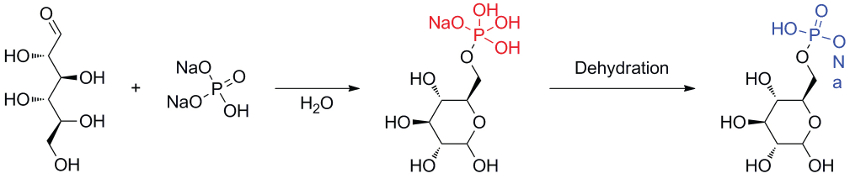
The phosphoranes were characterized using high-resolution electrospray-ionization mass spectroscopy (ESI-MS). The team also demonstrated the dehydration of phosphoranes to phosphates using tandem mass spectrometry (pictured above, second step).
- Characterization of Phosphoranes Obtained via the Spontaneous Carbonyl-Catalyzed Phosphorylation of Monosaccharides and Polyols In Aqueous Media,
Maria Perez-Ramirez, Isaac Agyekum, Barnabas Otoo,
ChemBioChem 2019.
https://doi.org/10.1002/cbic.201900328