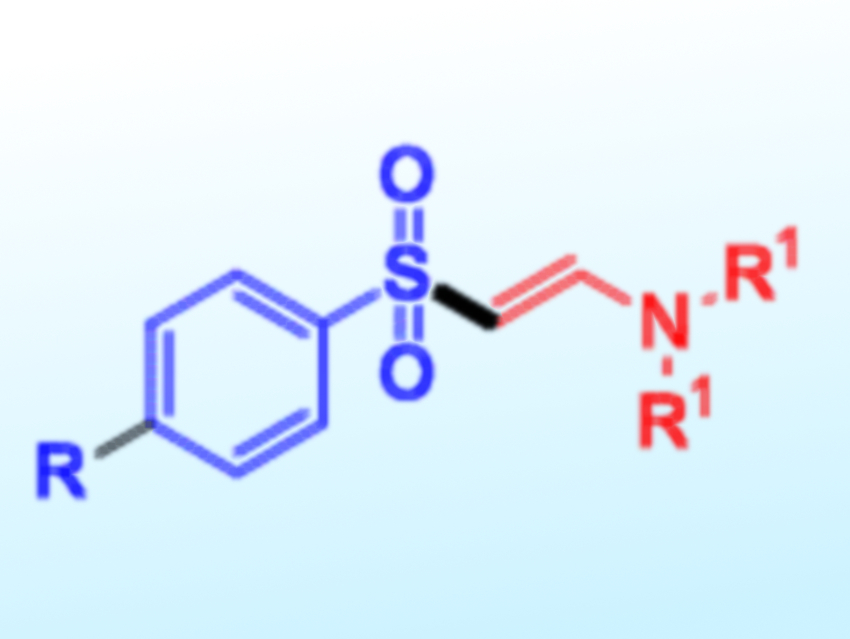
Electrochemical synthesis methods have several advantages in organic chemistry. They often proceed under mild conditions and are environmentally friendly. Sunwoo Lee, Chonnam National University, Gwangju, Republic of Korea, and colleagues have shown for the first time that β-amidovinyl sulfones can be obtained using electrochemistry.
β-Amidovinyl sulfones were synthesized via the reaction of arylsulfonyl hydrazides with tertiary amines under mild electrochemical conditions. The reactions were conducted in a solution of n-Bu4NBF4 and acetic acid in dimethyl sulfoxide in undivided cells with graphite and platinum electrodes. The reactions were performed under a constant current, at room temperature, and under air.
According to the team, both the tertiary amine and the arylsulfonyl hydrazide are activated at the anode. The amine is converted to an enamine intermediate and the arylsulfonyl hydrazide is transformed into an arylsulfonyl radical. the arylsulfonyl radical reacts with the enamine to provide a radical intermediate, which is oxidized and deprotonated to give the product. The desired β-amidovinyl sulfones were obtained in moderate to good yields.
- Electrochemical Coupling of Arylsulfonyl Hydrazides and Tertiary Amines for the Synthesis of β-Amidovinyl Sulfones,
Han-sung Kim, Sunwoo Lee,
Eur. J. Org. Chem. 2019.
https://doi.org/10.1002/ejoc.201901277