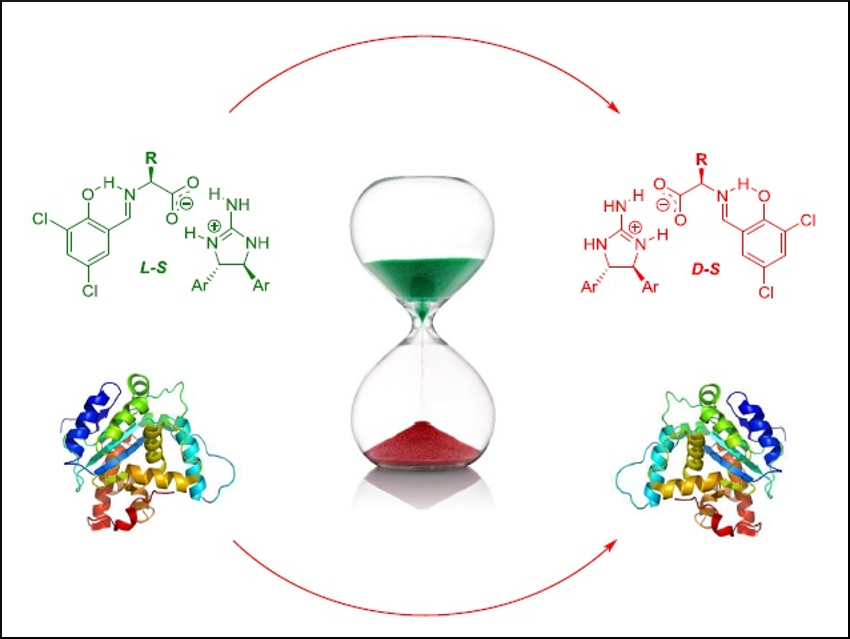
Amino acids are fundamental building blocks of life. Nearly all living organisms contain structures that are built from L-amino acids, while their mirror-images, the D-configured congeners, are unnatural. D-amino acids have a variety of uses in the pharmaceutical industry and biotechnology, most notably as building blocks of antibiotics. However, their widespread use is hampered by their cost and limited availability. Their synthesis usually involves multi-step procedures.
Jik Chin, University of Toronto, Canada, and colleagues have developed a simple one-step protocol with recyclable auxiliaries that transforms L-amino acids to D-amino acids (or vice-versa) by using differences in solubility between intermediate diastereomeric salts. The team calls the method solubility-induced diastereomer transformation (SIDT).
The intermediate diastereomeric salt was prepared by adding 3,5‐dichlorosalicylaldehyde and a chiral guanidine to a solution of the L-amino acid to create an imino acid salt. This salt rapidly switches between diastereomers. The less soluble diastereomer precipitates cleanly and is then hydrolyzed to give the enantiopure D-amino acid. The auxiliaries can be recovered for further cycles. Throughout the process, only filtration is needed for purification. The proposed method is potentially scalable and generally applicable for a wide variety of amino acids.
- Hydrogen Bond Assisted L to D Conversion of α-Amino Acids,
Rui Fu, Soon Mog So, Alan J. Lough, Jik Chin,
Angew. Chem. Int. Ed. 2020.
https://doi.org/10.1002/anie.201914797