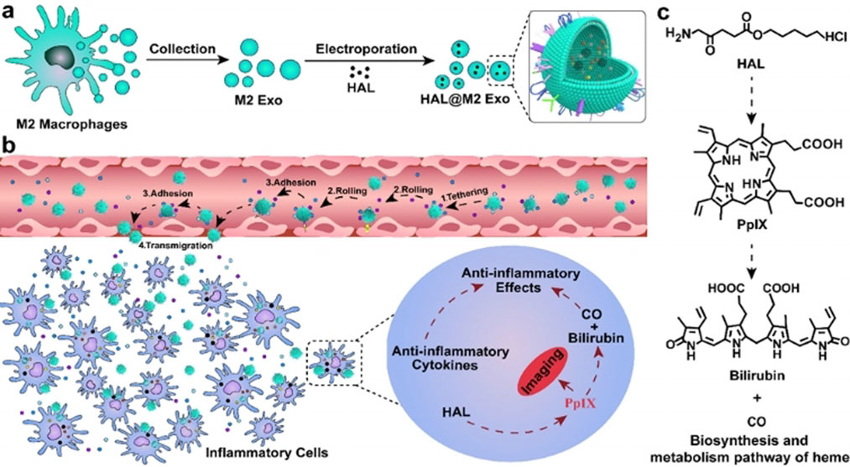
Exosomes are extracellular vesicles, i.e., small spherical compartments surrounded by a membrane. They are produced by most eukaryotic cells. Recently, exosome-based drug delivery systems have attracted research attention. However, exosomes that can help to target and treat inflammation are rare. Inflammation is, for example, an issue in atherosclerosis—a major contributor to heart disease.
Hai-Yan Xie, Beijing Institute of Technology, China, Kanyi Pu, Nanyang Technological University, Singapore, and colleagues have designed molecularly engineered exosomes derived from M2 macrophages (a type of white blood cell) that can target inflammation and help to treat it. The team introduced hexyl 5-aminolevulinate hydrochloride (HAL) into the M2 macrophage-derived exosomes (M2 Exo) through electroporation. Electroporation is a technique in which an electrical field is applied to cells in order to increase the permeability of the cell membrane and allow molecules to pass through.
The engineered exosomes can actively target inflammation sites. Inside the target cells, HAL initiates the biosynthesis of heme and leads to the production of two anti‐inflammatory compounds, carbon monoxide (CO) and bilirubin (pictured below). These compounds add to the effect of the anti-inflammatory cytokines contained in M2 Exo. Cytokines are small proteins produced, e.g., by the immune system.
In a mouse model, more than 75 % of atherosclerosis plaques could be alleviated using the engineered exosomes. Additionally, the protoporphyrin IX (PpIX) intermediate (pictured below) in the heme biosynthesis pathway enables fluorescence imaging and, thus, the tracking of atherosclerosis.
- Molecularly Engineered Macrophage-Derived Exosomes with Inflammation Tropism and Intrinsic Heme Biosynthesis for Atherosclerosis Treatment,
Guanghao Wu, Jinfeng Zhang, Qianru Zhao, Wanru Zhuang, Jingjing Ding, Chi Zhang, Haijun Gao, Dai-Wen Pang, Kanyi Pu, Hai-Yan Xie,
Angew. Chem. Int. Ed. 2020.
https://doi.org/10.1002/anie.201913700




