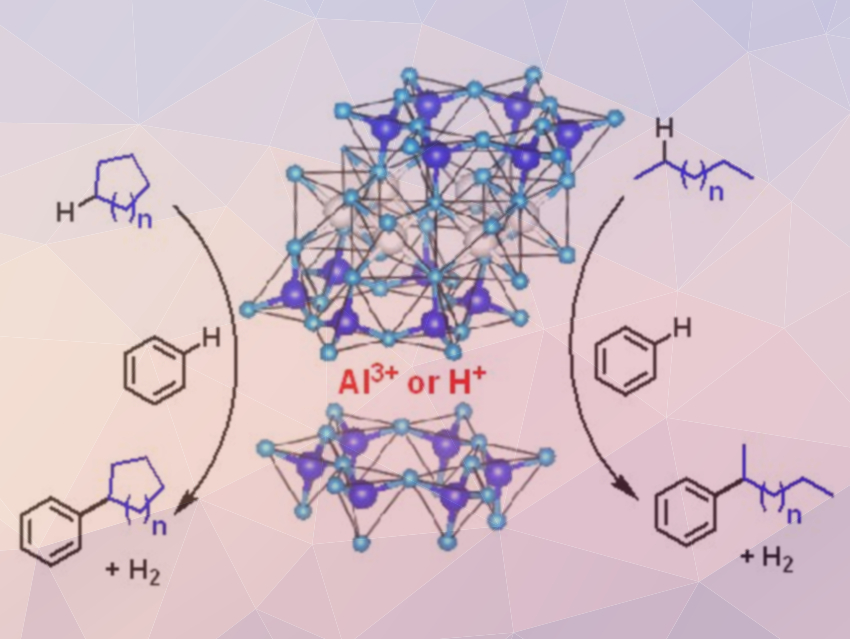
Alkylbenzenes are widely used as synthetic intermediates and as raw materials for the production of, e.g., synthetic detergents or organic solvents. Usually, they are obtained via a Friedel‐Crafts reaction between alkyl halides and benzene. However, organohalides are not environmentally benign and, thus, the direct alkylation of benzene with alkanes would be preferable. Direct reactions of benzene with propane and ethane have been reported using metal‐modified zeolites as catalysts at temperatures of 300–500 °C.
Ken Motokura, Tokyo Institute of Technology, Japan, and Japan Science and Technology Agency (JST), Saitama, and colleagues have found that the direct benzene alkylation with alkanes can be achieved at lower temperatures using montmorillonites as noble-metal-free solid acid catalysts. Montmorillonites are phyllosilicate minerals that can be used in a variety of catalytic processes. The team prepared hydrogen- aluminium-, and gallium‐exchanged montmorillonite from sodium‐type montmorillonite using an ion‐exchange procedure. To test these variants, the direct alkylation of benzene with n-heptane was performed at 150 °C.
Aluminum-exchanged montmorillonite showed the highest yield of the desired alkylated products. Under optimized reaction conditions, a benzene conversion of 2.6 % with a 73 % selectivity toward heptylbenzenes was achieved. The catalyst system was used with other linear and cyclic alkanes and gave the corresponding alkylated products with good selectivities. This montmorillonite-catalyzed direct alkylation system could contribute to environmentally benign chemical production processes.
- Direct Alkylation of Benzene at Lower Temperatures in the Liquid Phase: Catalysis by Montmorillonites as Noble‐Metal‐Free Solid Acids,
Moe Takabatake, Masayuki Nambo, Yuichi Manaka, Ken Motokura,
ChemPlusChem 2020.
https://doi.org/10.1002/cplu.202000090