Polycyclic aromatic hydrocarbons (PAHs) are a class of π-conjugated molecules with potential applications in optoelectronic materials and devices. The properties of such devices are determined by the molecular structures of their PAH constituents. Thus, methods for the rapid and atomically precise synthesis of PAHs are useful.
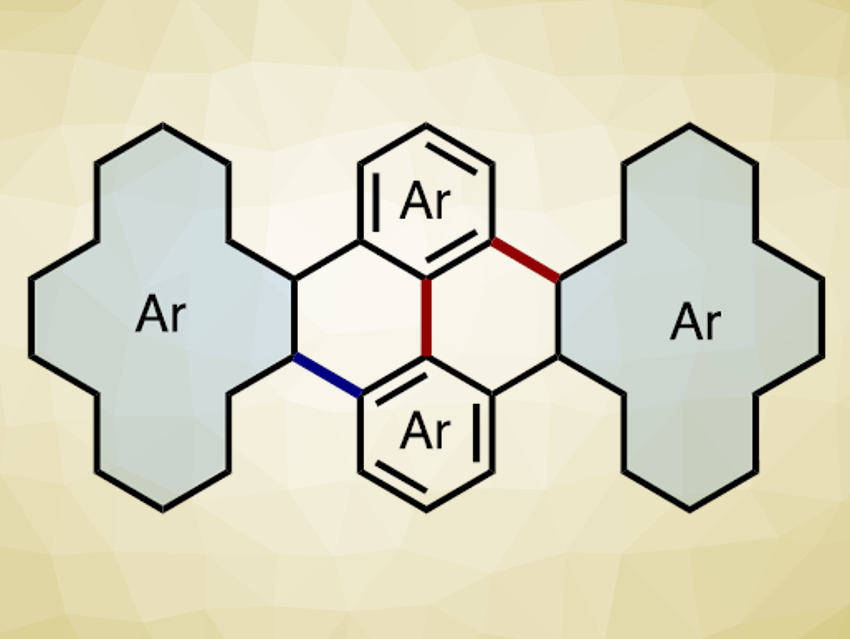
Kenichiro Itami, Kei Murakami, Nagoya University, Japan, and colleagues have developed a method for the synthesis of PAHs from simple phenol derivatives (pictured below). The team first used a palladium-catalyzed annulative dimerization of phenylene triflate to form a partially fused PAH. PdCl2 was used as a catalyst, tributylphosphine as a ligand, K2CO3 as a base, and pivalic acid as an additive. Then, an FeCl3-mediated Scholl reaction (a type of arene coupling) gave the desired fully fused PAH.
.jpg)
Using this approach, the researchers dimerized various substrates, such as tetraphenylene and binaphthalene triflates. The reaction proceeds regioselectively when phenanthrene- or pyrene-type substrates are used. According to the team, this strategy could simplify the preparation of PAHs compared with conventional synthetic methods.
- Synthesis of Polybenzoacenes: Annulative Dimerization of Phenylene Triflate by Twofold C−H Activation,
Mizuho Uryu, Taito Hiraga, Yoshito Koga, Yutaro Saito, Kei Murakami, Kenichiro Itami,
Angew. Chem. Int. Ed. 2020.
https://doi.org/10.1002/anie.202001211