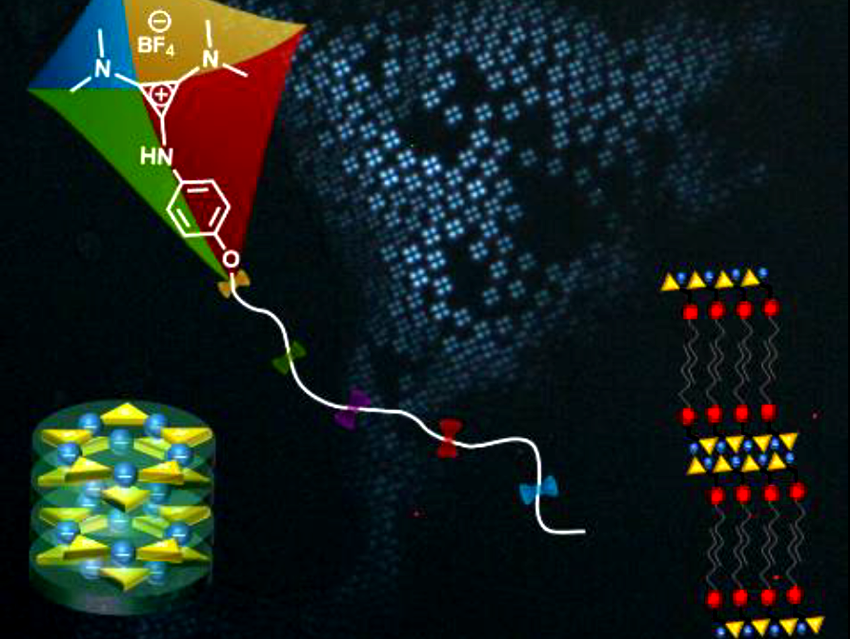
Cyclopropenium salts are three-membered π-systems and they are the smallest members of the Hückel aromatic family. These salts possess useful electronic and physical properties because they combine aromatic stability with ring strain and they can be tailored with substituents. Consequently, they are applied widely in applications such as organocatalysis and redox-active polymers. However, self-assembly of cyclopropenium compounds into liquid crystalline phases has not yet been reported.
Jeffrey Bandar, Colorado State University, USA, Tristan Lambert, Cornell and Columbia Universities, both New York, USA, Sabine Laschat, University of Stuttgart, Germany, and colleagues, have discovered that ionic liquid crystals (ILCs) can be obtained from aminocyclopropenium derivatives. The team combined 4-alkoxyanilines with cyclopropenium chloride in the presence of lithium chloride and Hünig’s base. After chromatographic purification and salt metathesis, cyclopropenium tetrafluoroborates were obtained.
The molten organic salts self-assemble into mesomorphic liquid crystalline phases—that is, phases between a solid and a liquid. Columnar or lamellar phase types can be tailored by the size of the headgroup and hydrophobic part of the aminocyclopropenium salts. The team discovered that segregation of immiscible parts of the complex, electrostatic interactions, and the volume of the head group and hydrophobic parts play a key role in mesophase stability.
- Self-Assembly of Aminocyclopropenium Salts: En Route to Deltic Ionic Liquid Crystals,
Juri Litterscheidt, Jeffrey S. Bandar, Max Ebert, Robert Forschner, Korinna Bader, Tristan H. Lambert, Wolfgang Frey, Andrea Bühlmeyer, Marcus Brändle, Finn Schulz, Sabine Laschat,
Angewandte Chemie International Edition 2020.
https://doi.org/10.1002/anie.202000824


![A Path to Substituted Bicyclo[2.1.1]hexanones](https://www.chemistryviews.org/wp-content/uploads/2024/10/1substitutedbicyclo211hexan2ones_2024-125x94.png)
