The detection of nucleotides such as adenosine triphosphate (ATP) is useful for a variety of applications in biology and medicine. One possible is approach is the use of artificial receptors that show a ratiometric fluorescent response upon nucleotide recognition, i.e., an output signal that is directly proportional to the input concentration.
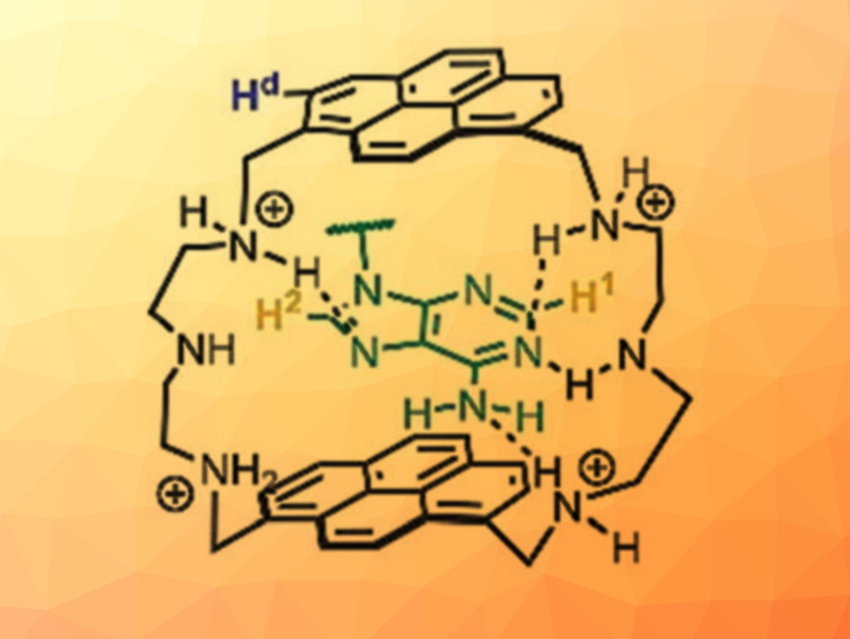
Evgeny A. Kataev, University of Erlangen-Nürnberg, Erlangen, Germany, and colleagues have developed pyrene-based macrocycles with a bellows-type sensing mechanism (pictured below) for the detection of nucleotides in water. The team synthesized four different macrocycles via condensation reactions of 2,2’‐diaminodiethylamine or 2‐(2‐aminoethoxy)ethylamine with 1,6‐ or 1,8-pyrenedicarboxaldehyde. The sensing mechanism is based on the encapsulation of a nucleobase between the two pyrene units, which moves them apart—like a bellows that expands. This affects the fluorescent properties of the molecule.
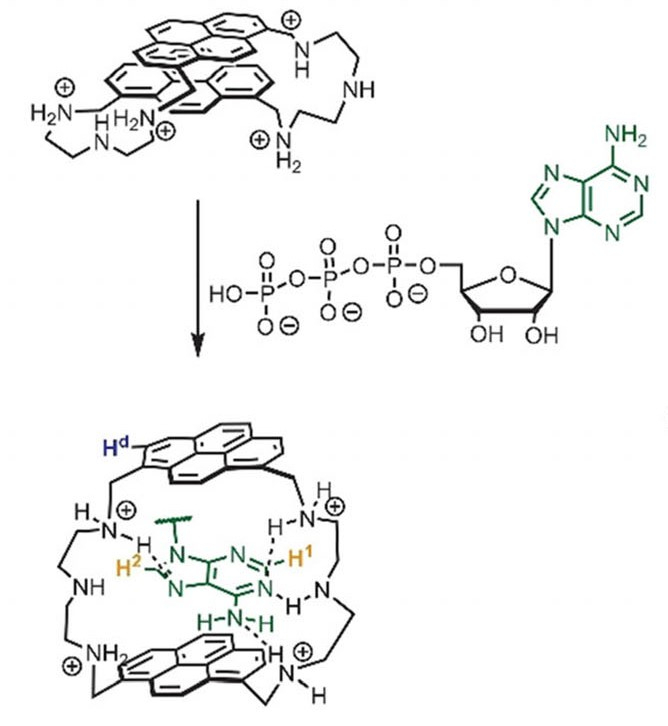
The 1,8‐substituted pyrene‐based cyclophane with the 2,2’‐diaminodiethylamine spacer showed the best selectivity for ATP. The high selectivity was explained by the combination of π–π stacking pyrene–adenine interactions with hydrogen bonding in the receptor–nucleotide complex (pictured). The presented bellows-type sensing mechanism is new and could be useful to construct other receptors.
- Ratiometric Detection of ATP by Fluorescent Cyclophanes with Bellows‐Type Sensing Mechanism,
Aleksandr M. Agafontsev, Tatiana A. Shumilova, Aleksandr S. Oshchepkov, Frank Hampel, Evgeny A. Kataev,
Chem. Eur. J. 2020.
https://doi.org/10.1002/chem.202001523