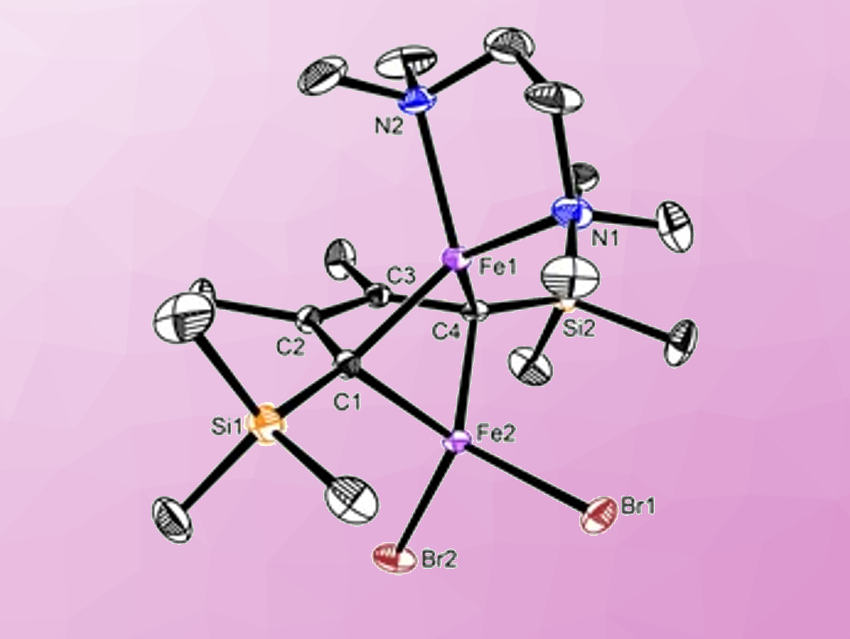
Aromaticity is one of the fundamental concepts in chemistry. For most aromatics, electron delocalization in the conjugated ring is considered to be achieved via π-type orbital overlap (“side-by-side”). However, σ-type orbital overlap in a “head-to-head” fashion is rarely proposed even in nonplanar metalla-aromatic conjugated systems.
Zhenfeng Xi and colleagues, Peking University, Beijing, China, have introduced two iron centers as a “superatom” into the butadienyl metallacyclic system. The team synthesized butadienyl diiron complexes with a more significant nonplanarity than previously reported in nonplanar aromatics. They used X-ray structural analysis and theoretical approaches to study the electronic structure and aromaticity of the new compounds.
In the nonplanar ferracycle, electron delocalization, as the main origin of aromaticity, results from σ-type overlap between the ligand p and metal d orbitals. In contrast to normal systems with π-type orbital overlap, these nonplanar metalla-aromatics presents a typical example of an unusual way to achieve electron delocalization on conjugated rings.
- Butadienyl Diiron Complexes: Nonplanar Metalla‐Aromatics Involving σ‐Type Orbital Overlap,
Chao Yu, Mingdong Zhong, Yongliang Zhang, Junnian Wei, Wangyang Ma, Wen-Xiong Zhang, Shengfa Ye, Zhenfeng Xi,
Angew. Chem. Int. Ed. 2020.
https://doi.org/10.1002/anie.202008986