Oxindoles and oxetanes (pictured below) are structures found in a number of bioactive molecules. Synthetic strategies that simultaneously functionalize oxindoles and install an oxetane ring can be used to generate compounds with potential biological activity. However, this is challenging due to the high ring strain of oxetane and to the low reactivity at the oxindole’s C2 position.
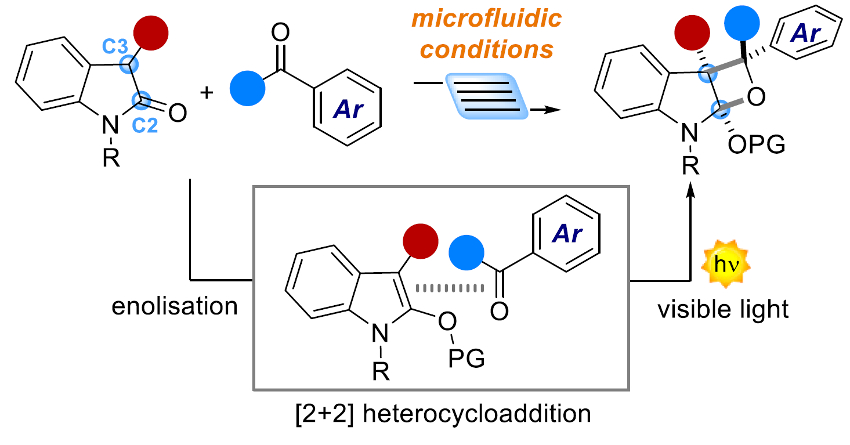
Luca Dell’Amico, University of Padova, Italy, and colleagues have used a visible-light-driven [2+2] cycloaddition to synthesize new fused oxindole–oxetane compounds (pictured below). This light‐driven [2+2] heterocycloaddition process is known as the Paternò–Büchi (PB) reaction. The researchers used simple aromatic ketones and a variety of different oxindoles as substrates. The oxindoles were first converted to the corresponding enol ethers. These were then reacted with ketones in a microfluidic photoreactor (MFP) setup under a 405 nm LED light. The ketone absorbs light and reaches its excited state, which then reacts with the enol form of the oxindole to give the desired products.
.jpg)
The process is mild and highly selective. The team showed the generality of the microfluidic approach via the successful conversion of a variety of substrates (23 examples). They achieved yields up to >98 % and diastereomeric ratios up to >20:1. The process can be easily scaled up, and the researchers obtained up to 1.18 g of functionalized oxindole product.
- Microfluidic Visible‐Light Paternò‐Büchi Reaction of Oxindole Enol Ethers,
Pietro Franceschi, Javier Mateos, Alberto Vega Peñaloza, Luca Dell’Amico,
Eur. J. Org. Chem. 2020.
https://doi.org/10.1002/ejoc.202001057