Breast milk remains the “gold standard” of baby nutrition. In addition to water, lactose (which delivers energy), fats, and proteins, breast milk contains a number of other important components needed for the healthy development of an infant. One of these classes of biologically active substances is comprised of the human milk oligosaccharides (HMOs). In this part, we will take a closer look at the effects of HMOs.
6. Metabolism of HMOs [29]
The metabolism of the roughly 200 known HMO compounds is complex, and our knowledge about the metabolism of individual compounds remains very limited. The most important findings are summarized below.
In the acidic environment of the infant stomach, there is little degradation of HMOs. Even the pancreatic enzymes present in the small intestine largely leave HMOs undigested. It is in the large intestine that HMOs serve as substrates for bacterial hydrolysis and fermentation. They provide intestinal bacteria (bifidobacteria) with energy, and the resulting metabolic products then act as the nutritional foundation of the mucosal lining of the large intestine.
This cultivates microbial intestinal flora that are strongly determined by the available nutrients. In infants, these come from breast milk or formula. Because the composition of breast milk is different for every individual, the intestinal microbiomes of infants are correspondingly varied. (see Chapter 2.1).
Only about 1 % of HMOs are absorbed into the bloodstream from the small intestine every day. That is about 100 mg HMO for a typical intake of 10 g HMO/day. The blood transports these to the organs, including the brain. This can be detected through isotope labeling with 13C during biosynthesis within the mammary glands and subsequent analysis of the urine and blood plasma of the child. Another portion of the HMOs remain unchanged in the intestine and pass into the infants’ stool.
7. Functions of HMOs
Because of the structural complexity of the many different HMOs in breast milk, it is a major challenge to determine which HMO molecules or combination of HMO molecules are responsible for which physiological effects. Scientific studies of HMOs in cell cultures (in vitro), as well as animal and human studies (in vivo) demonstrate that HMOs support and influence the development and immune systems of infants. This partly results from their prebiotic effect: during the first few months, certain microorganisms, such as bifidobacteria and lactic acid bacteria, are fortified in the digestive tracts of nursing infants by HMOs from breast milk. These bacteria have special enzymes that break down HMOs, some with pronounced substrate specificity [4,30].
Compared with “bottle” babies, nursing infants have a lower probability of catching a variety of diseases caused by viruses, bacteria, fungi, and parasites. This can be traced back to the antimicrobial effect of fucosylated HMOs (like 2′-FL (4) or LNFP I (14)), as well as the antiadhesive effect of sialylated HMOs (like 3′-SL (16) or 6′-SL (17) [4,9,30,31].
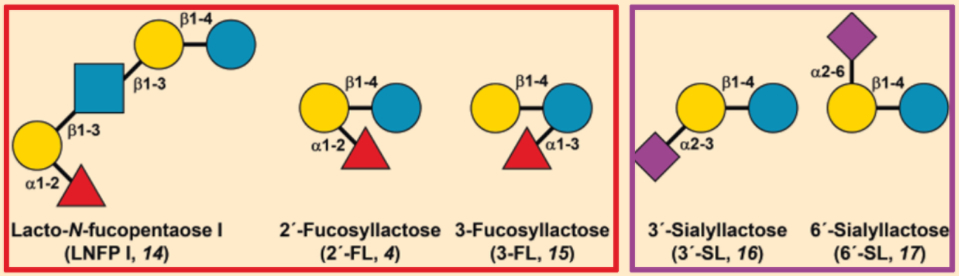
For many infections, adhesion of the pathogen to the intestinal mucosa is necessary. The specificity of this process depends on the macromolecular structures of the intestinal lining, which are entirely or partially made of sugars. Many HMOs resemble this receptor structure and can act as “decoys” for the pathogens. The presence of HMOs in the intestine can, thus, hinder adhesion of the pathogen to the intestinal mucosa, preventing infection. The pathogens are then evacuated from the body along with the HMOs (see Fig. 6).
.jpg) |
|
Figure 6. Pathogenic bacterium binds to “decoy” HMO. |
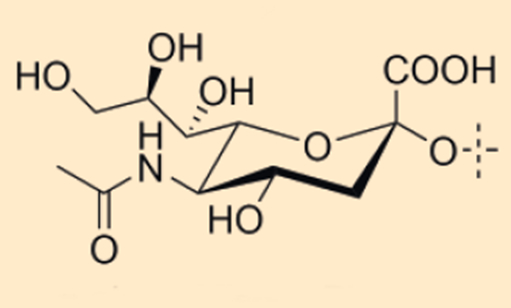
N-Acetyl-D-Neuraminic acid (13), an important representative of the sialic acids, plays a significant role in the brain development of newborns (see Fig. 7). This molecule is an essential building block in the synthesis of glycolipids and proteins. Human breast milk is primarily rich in sialylated HMOs in the first few hours after birth—fitting for the “developmental leap” made by the brain within this time frame. Studies with rats have shown that the ingestion of sialyllactoses (3′-SL (16), 6′-SL (17)) increases the amount of sialic acid in the brain [4].
 |
|
Figure 7. N-Acetyl-D-neuraminic acid (13). |
Interestingly, elephant milk is very similar to human milk with regard to the amounts of sialylated oligosaccharides. This may not be a coincidence—elephants are known for their outstanding memory (“a memory that never forgets”) and intelligence [32]. It is no surprise that sialylated HMOs are a focus for producers of formula.
8. Additional Research
In addition to the broad area of infant nutrition, HMOs are also being studied as building blocks for the development of antimicrobial agents [31,33] or as nutritional supplements for those with chronic bowel inflammation. More extensive studies about the absorption, metabolism, and physiological function of HMOs in infants and young children are needed to determine which HMOs, in what range of concentrations, and in which combinations with other HMOs lead to particular effects.
References
[29] S. Moukarzel, L. Bode, Human Milk Oligosaccharides and the Preterm Infant, Clin. Perinatol. 2017, 44, 193–207. https://doi.org/10.1016/j.clp.2016.11.014
[30] J. Brand Miller, P. McVeagh, Human milk oligosaccharides: 130 reasons to breast-feed, Br. J. Nutr. 2007, 82, 333–335. https://doi.org/10.1017/s0007114599001567
[31] K. M. Craft, S. D. Townsend, The Human Milk Glycome as a Defense Against Infectious Diseases: Rationale, Challenges, and Opportunities, ACS Infect. Dis. 2017, 4, 77–83. https://doi.org/10.1021/acsinfecdis.7b00209
[32] C. Kunz et al., Lactose-derived oligosaccharides in the milk of elephants: comparison with human milk, Br. J. Nutr. 2010, 82, 391–399. https://doi.org/10.1017/S0007114599001798
[33] D. L. Ackerman et al., Human Milk Oligosaccharides Exhibit Antimicrobial and Antibiofilm Properties against Group B Streptococcus, ACS Infect. Dis. 2017, 3, 595–605. https://doi.org/10.1021/acsinfecdis.7b00064
The Authors
 Michael Breuer, born in 1965, studied biology at the University of Bonn, Germany. He completed a research fellowship with H. G. Floss at the University of Washington, USA, in 1992–1993. He received his doctorate from the University of Bonn in 1995 for research under the supervision of E. Leistner into ansamacrolide biosynthesis in actinomycetes. Michael Breuer became Laboratory Manager in the Main Laboratory of BASF in Ludwigshafen, Germany, in 1996.
Michael Breuer, born in 1965, studied biology at the University of Bonn, Germany. He completed a research fellowship with H. G. Floss at the University of Washington, USA, in 1992–1993. He received his doctorate from the University of Bonn in 1995 for research under the supervision of E. Leistner into ansamacrolide biosynthesis in actinomycetes. Michael Breuer became Laboratory Manager in the Main Laboratory of BASF in Ludwigshafen, Germany, in 1996.
 Melanie Weingarten, born in 1978, studied chemistry at RWTH Aachen University, Germany, as a fellow of the German Academic Scholarship Foundation. She completed her undergraduate work in the field of synthesis and medicinal chemistry in 2003 at the University of California, Berkeley, USA, in the research group of J. Ellman and D. Enders. In 2006, she completed her doctoral degree on the subject of “Iron-catalyzed cross-coupling and total synthesis of myxovirescin A1” with A. Fürstner at the Max-Planck-Institute for Coal Research in Mülheim and der Ruhr, Germany. After a post-doc in the field of biochemistry and microbiology with B. Imperiali at the Massachusetts Institute of Technology (MIT), Cambridge, USA, Melanie Weingarten began work as a Laboratory Manager at BASF in Ludwigshafen, Germany, in the White Biotechnology Research division. She is currently working in the area of synthesis and homogeneous catalysis .
Melanie Weingarten, born in 1978, studied chemistry at RWTH Aachen University, Germany, as a fellow of the German Academic Scholarship Foundation. She completed her undergraduate work in the field of synthesis and medicinal chemistry in 2003 at the University of California, Berkeley, USA, in the research group of J. Ellman and D. Enders. In 2006, she completed her doctoral degree on the subject of “Iron-catalyzed cross-coupling and total synthesis of myxovirescin A1” with A. Fürstner at the Max-Planck-Institute for Coal Research in Mülheim and der Ruhr, Germany. After a post-doc in the field of biochemistry and microbiology with B. Imperiali at the Massachusetts Institute of Technology (MIT), Cambridge, USA, Melanie Weingarten began work as a Laboratory Manager at BASF in Ludwigshafen, Germany, in the White Biotechnology Research division. She is currently working in the area of synthesis and homogeneous catalysis .
The article has been published in German as:
- Mehr als 200 gute Gründe für das Stillen,
Michael Breuer, Melanie Weingarten,
Chem. unserer Zeit 2019, 53, 94–104.
https://doi.org/10.1002/ciuz.201900030
and was translated by Caroll Pohl-Ferry.
Nursing, Gut Bacteria, and the Immune System – Part 1
Which components are found in breast milk?
Nursing, Gut Bacteria, and the Immune System – Part 2
What are human milk oligosaccharides (HMO)?
Nursing, Gut Bacteria, and the Immune System – Part 3
How are human milk oligosaccharides (HMO) synthesized?
See similar articles published on ChemistryViews.org



