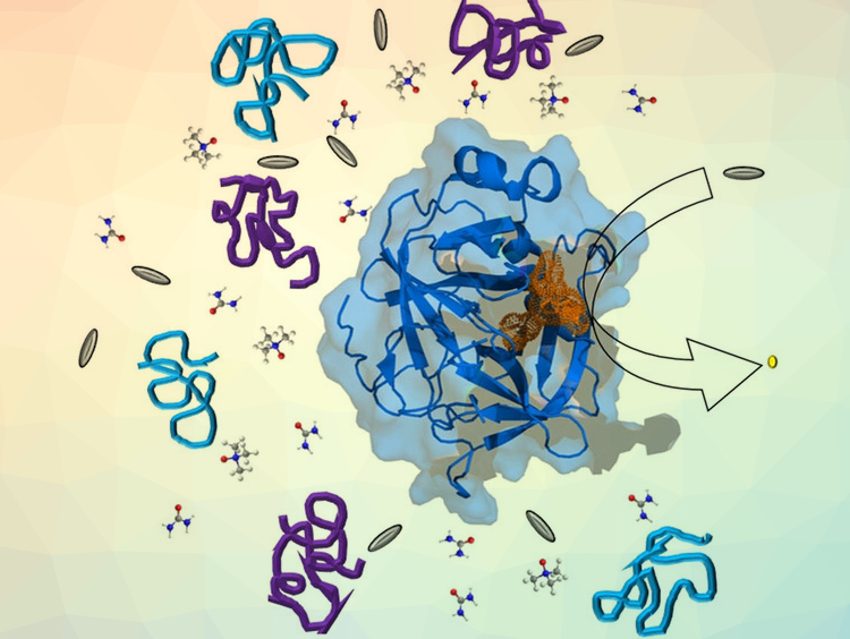
Understanding the parameters that influence enzymatic activity is important to control and optimize enzyme-driven reactions. For example, at a cellular level, the effects of macromolecular crowding agents and osmolytes must be considered. Osmolytes are low-molecular-weight organic compounds that influence the properties of biological fluids. Macromolecular crowding agents such as proteins can alter the properties of a solution when they are present in high concentrations, as is often the case in living cells.
Michel W. Jaworek and Roland Winter, TU Dortmund University, Germany, have studied the combined effects of macromolecular crowding, co-solutes, and high pressure on the activity of the model enzyme α-chymotrypsin (α-CT). The enzymatic activity was investigated using the α-CT-catalyzed hydrolysis of N-succinyl-Ala-Ala-Pro-Phe-p-nitroanilide (SAAPPpNA) as a model reaction. Polyethylene glycol (PEG) and dextran were used as the crowding agents, urea and trimethylamine-N-oxide (TMAO) as co-solutes. In addition, hydrostatic pressures between atmospheric pressure and 2 kbar were applied.
The team found that crowding agents and co-solutes can have diverse effects on the enzymatic reaction that are difficult to predict. In all cases, increasing the pressure increased the turnover number of the reaction. The catalytic efficiency of the reaction was highest in the presence of the osmolyte TMAO, with higher pressures increasing this effect. Urea, in contrast, has a negative effect on the catalytic efficiency. PEG and dextran, two crowding agents of a different nature, lead to very similar catalytic efficiencies.
According to the researchers, the work may be relevant for enzymatic industrial processes, where enhancing enzyme activity could, for example, optimize yields, increase turnover rates, and improve cost-effectiveness. The study may also help to gain insight into cellular processes in organisms found in extreme environments such as the deep sea.
- Exploring Enzymatic Activity in Multiparameter Space: Cosolvents, Macromolecular Crowders and Pressure,
Michel W. Jaworek, Roland Winter,
ChemSystemsChem 2020.
https://doi.org/10.1002/syst.202000029