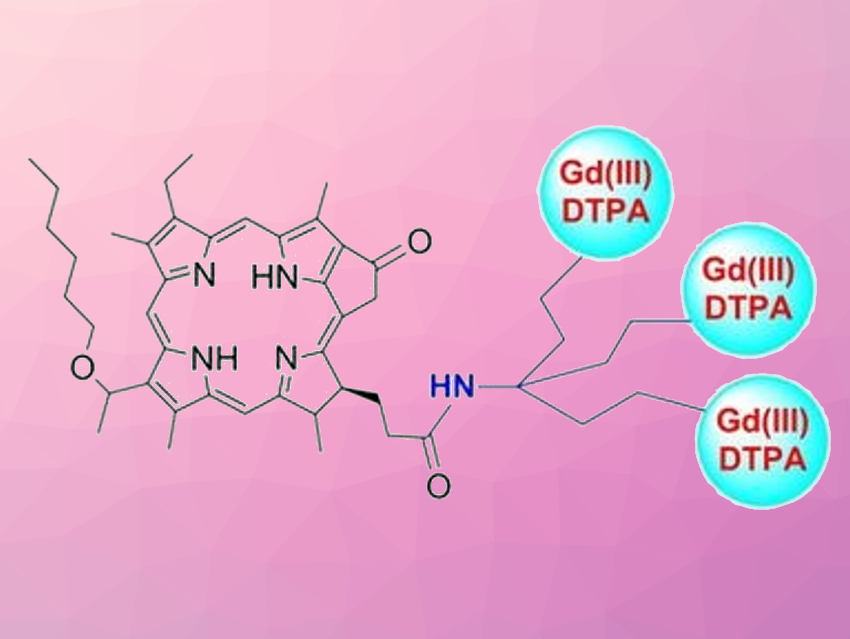
Magnetic resonance imaging (MRI) can be very useful in cancer diagnosis. However, MRI has poor sensitivity and high costs. The additional use of optical fluorescence imaging (OFI), which combines fast acquisition, high sensitivity, and low cost, can help to provide a better view of the disease. Bifunctional agents for tumor imaging and photodynamic therapy (PDT) can be particularly useful in this context. The second-generation photosensitizer 3-(1′-hexyloxyethyl)-3-devinylpyropheophorbide-a (HPPH), for example, is currently undergoing human clinical trials. Evaluating the impact of structural modifications on the properties of HPPH could lead to even better multifunctional agents.
Joseph A. Spernyak, Ravindra K. Pandey, Roswell Park Comprehensive Cancer Center, Buffalo, NY, USA, and colleagues have bound HPPH to gadolinium chelate complexes, i.e., Gd(III)-aminobenzyl-DTPA (Gd(III) DTPA, DTPA = diethylenetriaminepentacetate, example conjugate pictured) or Gd(III)-aminoethylamido-DOTA (Gd(III) DOTA, DOTA = dodecane tetraacetic acid), at various positions. Gadolinium-containing compounds are often used as MRI contrast agents, and thus, such conjugates could combine this use with fluorescence imaging and photodynamic therapy. All conjugates were assessed for their efficacy in photodynamic therapy, their in vivo fluorescence, and their suitability as an MRI contrast agent.
The team found that the water‐soluble Gd(III)‐aminoethylamido‐DOTA linked at position‐17 of HPPH was the most promising agent. It showed good efficacy in photodynamic therapy, with a tumor cure rate of 70 % in a mouse model. The compound remained in a tumor for a long period of time (24–48 h) and provides the option of fluorescence‐guided cancer therapy. The team points out that pharmacokinetics, pharmacodynamics, and toxicological studies of HPPH-17-Gd(III) DOTA are needed before initiating human clinical trials.
- The Structures of Gd(III) Chelates Conjugated at the Periphery of 3‐(1’‐Hexyloxy)ethyl‐3‐devinylpyropheophorbide‐a (HPPH) Have a Significant Impact on the Imaging and Therapy of Cancer,
Shunqing Zhang, Ravindra R. Cheruku, Mykhaylo Dukh, Walter Tabaczynski, Nayan J. Patel, William H. White, Joseph R. Missert, Joseph A. Spernyak, Ravindra K. Pandey,
ChemMedChem 2020.
https://doi.org/10.1002/cmdc.202000449