The hydrogenation of multiple bonds is an important reaction in catalysis research and also industrially relevant. Usually, late transition metals are used in this type of reaction. However, complexes of the alkaline earth metals calcium and barium can also act as hydrogenation catalysts. The activity of this type of catalyst generally increases with the steric bulk of the ligands.
Sjoerd Harder and colleagues, University of Erlangen-Nürnberg, Erlangen, Germany, have found that barium metal with no ligands at all can be a highly active hydrogenation catalyst. Barium metal was activated by evaporation and co-condensation with heptane under high vacuum. The resulting black barium metal powder is a highly active hydrogenation catalyst for the reduction of a variety of unactivated, non-conjugated mono-, di-, and tri-substituted alkenes, tetraphenylethylene, benzene, different polycyclic aromatic hydrocarbons, aldimines, ketimines, and pyridine derivatives with H2 gas.
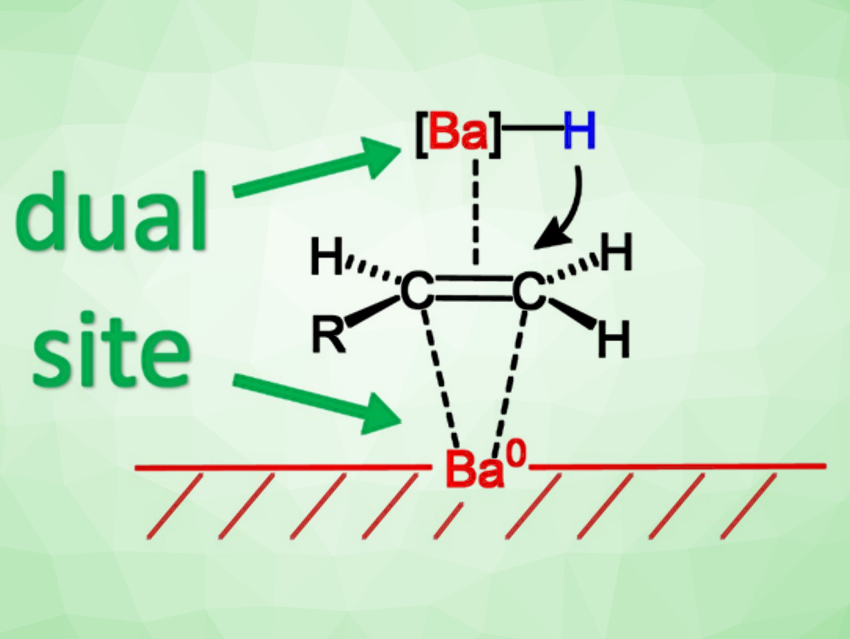
Metallic barium outperforms all known molecular group 2 metal hydrogenation catalysts. The team proposes a dual-site mechanism in which both barium metal and barium hydride formed in situ work together (pictured). Although the metal needs to be activated, using barium metal as a catalyst is a much simpler approach than preparing the previously known molecular barium catalysts with bulky ligands.
- Metallic Barium: A Versatile and Efficient Hydrogenation Catalyst,
Sjoerd Harder, Philipp Stegner, Christian Färber, Ulrich Zenneck, Christian Knüpfer, Jonathan Eyselein, Michael Wiesinger,
Angew. Chem. Int. Ed. 2020.
https://doi.org/10.1002/anie.202014326