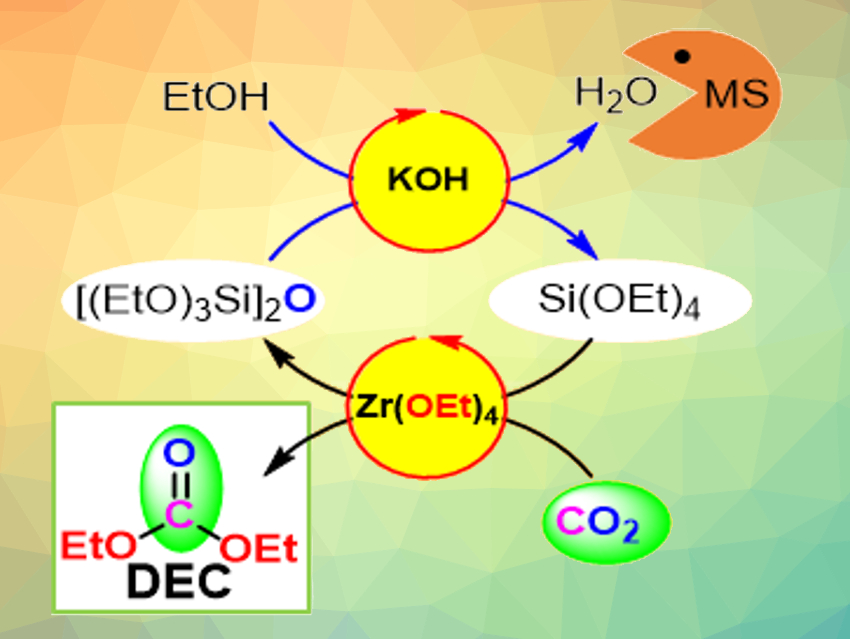
Catalytic transformations of CO2 into fine chemicals can be used to reduce the reliance on fossil resources and mitigate carbon emissions. The conversion of CO2 into diethyl carbonate (DEC), for example, is useful because of DEC’s applications in batteries and polymer synthesis. However, the direct DEC synthesis via a reaction of ethanol and CO2 is limited by its chemical equilibrium. Water is produced as a byproduct in this reaction and an effective combination of catalyst and dehydrating agent is necessary.
Jun-Chul Choi, Norihisa Fukaya, National Institute of Advanced Industrial Science and Technology (AIST), Tsukuba, Japan, and colleagues have developed a sustainable synthesis of DEC from CO2 using ethoxysilane derivatives and Zr(OEt)4 (pictured). The team used tetraethyl orthosilicate (TEOS) as a reactant to avoid the formation of water. Instead, a disiloxane byproduct is created, which can be converted back to TEOS with ethanol, using molecular sieves as a dehydrating agent. Zr(OEt)4 acts as the catalyst for DEC formation.
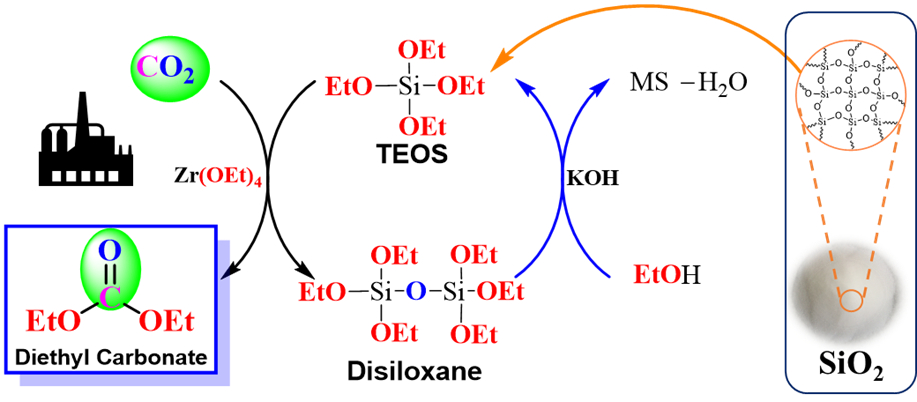
With this approach, the team achieved a record turnover number for DEC synthesis of 43. The regenerable disiloxane byproduct points to a new direction for the development of waste-free syntheses of DEC according to the researchers.
- Sustainable Catalytic Synthesis of Diethyl Carbonate,
Wahyu S. Putro, Akira Ikeda, Shinji Shigeyasu, Satoshi Hamura, Seiji Matsumoto, Vladimir Ya. Lee, Jun-Chul Choi, Norihisa Fukaya,
ChemSusChem 2020.
https://doi.org/10.1002/cssc.202002471