What is the connection between food and brain function? Can certain ingredients alter brain activity, influencing behavior and making us eat more? At the same time, can our brain function determines what and how much we consume?
1. Molecular Nutrition Physiology
Molecular nutrition physiology is concerned with the question of what effects food ingredients can have over specific bodily functions, above and beyond the provision of energy and nutrients. In contrast to other disciplines, it does not focus on the effects of different forms of nutrition or foods. Instead, the goal is to assign a specific physiological activity to a molecularly defined compound.
One important requirement for reaching this goal is interdisciplinary collaboration. The research association called Neurotrition thus involves scientists from fields that include food chemistry, food analysis, sensor technology, computational chemistry, physiology, clinical pharmacy, medicinal chemistry, neurobiology, and preclinical imaging, as well as clinical disciplines such as psychiatry, endocrinology, and neuroradiology. These researchers are working together with the goal of investigating the interactions between food ingredients and brain function. Food chemistry takes on a central role in this work, because it can be used to identify new neuroactive substances in foods, determine their concentrations in the food and after resorption in the human body, and examine their effects on different metabolic pathways.
The interactions between foods and brain function are bidirectional and manifold: On the one hand, foods influence our brain function; on the other, our brain function also influences how much and which foods we consume. Both processes can also depend on each other: Certain food ingredients lead to changes in the activity patterns of the brain. This eventually leads to behavioral changes that cause us to consume these foods more frequently and in larger amounts.
2. Food Ingredients Influence Our Brain Function
2.1. Coffee
Many of us have already discovered for ourselves that food can fundamentally influence brain function. At least, this is true for those who need a cup of coffee first thing in the morning before they can face the challenges of the day. The neurophysiological effect of coffee has been known for a long time.
The caffeine in coffee is an antagonist that blocks the A1 and A2A subtypes of the adenosine receptor in various regions of the brain. This causes the ligand that naturally binds to this site, adenosine, to be unable to bind. Adenosine is a “tiredness signal” in the brain, and its interaction with the adenosine receptors initiates sleepiness. This is how caffeine suppresses sleep, improves alertness, and can increase physical endurance [1,2].
There are indications of other effects of foods on brain function; however, the physiological mechanisms and molecular structures of the substances are rarely known. Some of these mechanisms were thus clarified as part of the Neurotrition project.
2.2. GABA Receptor
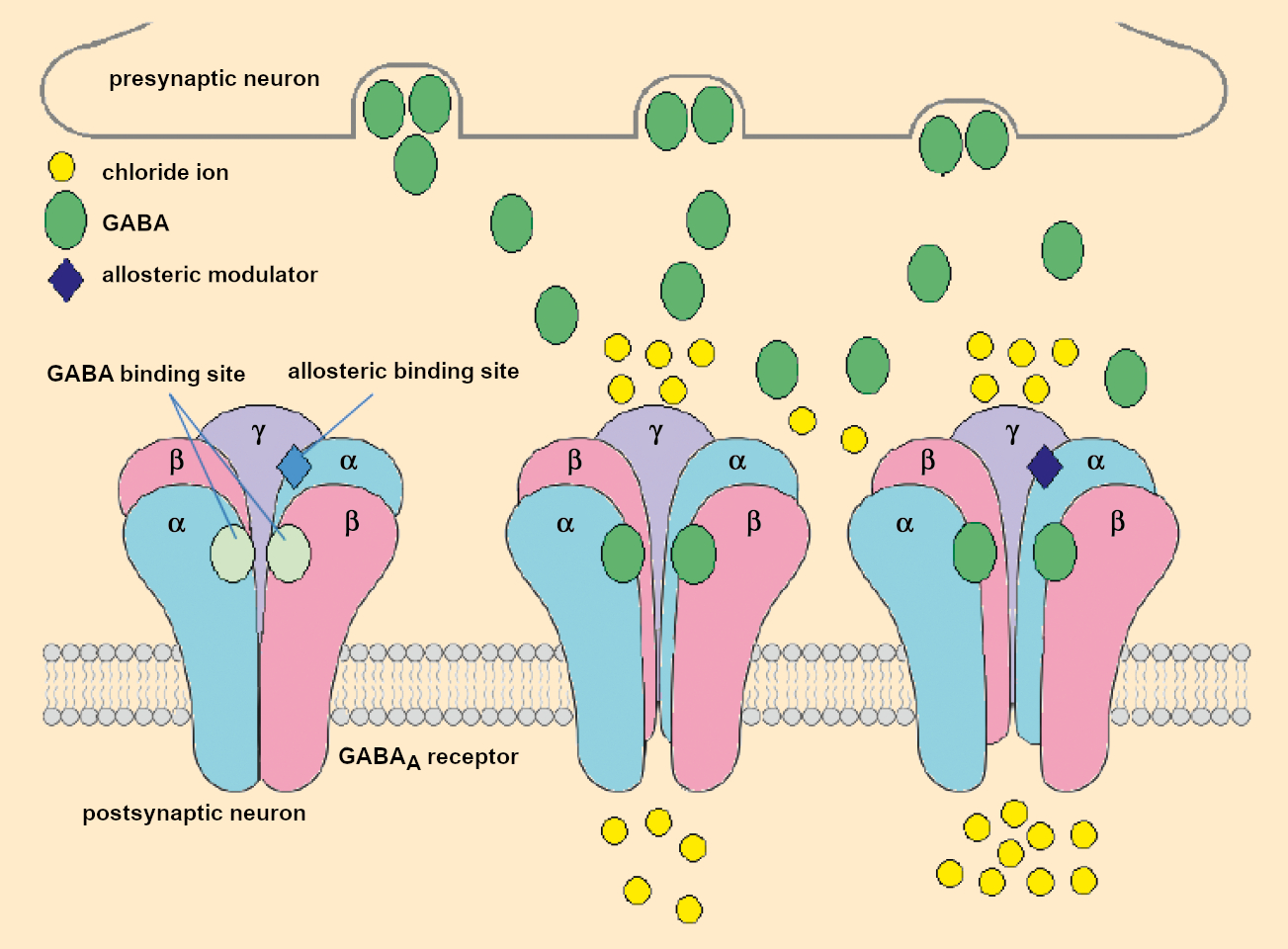
One example of this is that of foods which can modulate the GABA receptor. GABA receptors are transmembrane proteins in nerve cells. They specifically bind to the neurotransmitter γ-aminobutyric acid (GABA). Like the adenosine receptor, the GABA receptor—or more precisely, the GABAA receptor—is found in many areas of the brain. It is primarily involved in chemical signal transmission between two nerve cells in a synapse.
If the first nerve cell releases the messenger molecule γ-aminobutyric acid (GABA), it binds to GABAA receptors on the surface of the next nerve cell (see Fig. 1). The GABAA receptor is a ligand-gated ion channel. This means that a channel is opened in the receptor when the GABA ligand is bound to it. This allows for the influx of ions from the synapse into the cell interior. The channel is constructed to selectively allow only negatively charged ions. The influx of chloride ions increases the resting potential of the cell, which makes it less excitable.
 |
|
Figure 1. Schematic representation of the allosteric activation of the GABAA receptor. |
This is why substances that amplify this process often have a soporific, calming, or anxiolytic effect. Many sleeping pills, such as barbiturates or benzodiazepines (e.g., the drug Diazepam in the brand-name product Valium), take effect by way of the GABAA receptor. They often directly mimic the effect of GABA but react at a different location on the receptor. This allows GABA to bind more strongly, leaving the ion channel open longer (allosteric activation) [3]. Food ingredients can also take effect through the GABAA receptor. However, their effects are usually significantly milder than the pharmaceuticals.
3. The Search for Neuroactive Food Ingredients Using the Example of GABAergic Substances
3.1. Screening Process
There are a variety of possibilities for identifying neuroactive foods and food ingredients. The best means of first getting oriented is a screening process. This allows the largest possible number of extracts or ingredients to be tested for physiological activity. Because it is usually not initially known which extracts or substances are promising, the basic requirement for a successful screening process is the availability of a rapid biotest (bioassay, a test to determine the biological effect of a substance). This makes it possible to test many sample solutions in a manageable timeframe and maybe find the “needle in a haystack”.
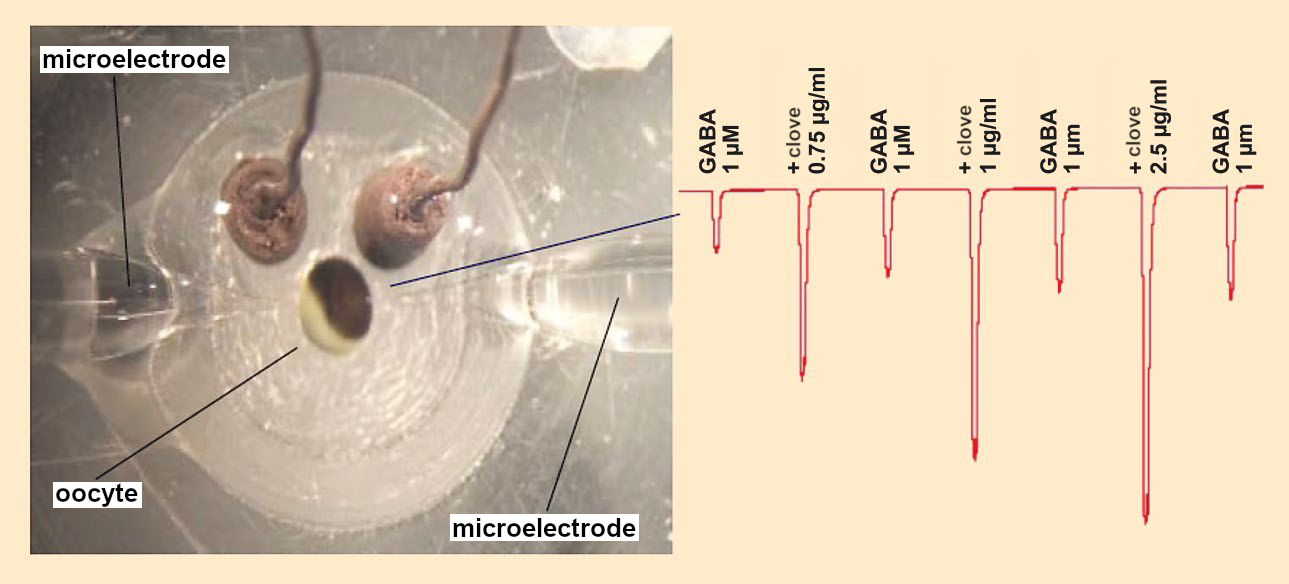
One biotest for the effects of foods on the GABA system is the two-electrode voltage clamp (TEVC) technique (see Fig. 2). This method uses frog eggs (oocytes from Xenopus laevis). The surfaces of these eggs contain practically no signal molecules but can be altered through molecular biology to specifically form the GABAA receptor. Two microelectrodes can then be introduced to a single egg cell in such a way that the electrical potential between the inside and outside can be measured. This makes it possible to measure the activation of ion channels.
 |
|
Figure 2. Measurement of the GABAA receptor-mediated chloride ions stream in oocytes by the two-electrode voltage clamp (TEVC) method. |
When the oocyte is rinsed with a GABA solution, the ion channel opens and chloride ions from the environment stream into the egg cell. This increases the negative charge in the interior and the change in voltage can be measured. When an active test substance is then added to the GABA solution, the chloride ion stream is intensified, which can be measured by an increase in the voltage change.
It is even possible to use TEVC to measure several substances in sequence. However, the measurement of a single substance takes (not including preparation of the oocytes) about 10 minutes. This makes TEVC impractical for high-throughput screening. The search for GABAergic, neuroactive substances is thus carried out with effect-directed screening.
In this method, foods are selected for which there are indications or individual reports that they may be calming, soporific, or anxiolytic, but whose mode of action and active ingredients are unknown. This should make the ratio of successful hits higher than for a random selection of test substances. By this method, it was demonstrated that extracts of various foods, such as cocoa, mountain tea (Sideritis), pepper, and hops can amplify the activity of GABA by up to a factor of four. The strongest effect was found for a clove extract (Syzygium aromaticum) [4]. This result is amazing in that no calming or sleep-inducing effect has been described for cloves. However, the local pain-relieving effect of cloves was clinically proven long ago.
This newest observation fits well with newer studies that ascribe GABA a significant role in the experience of pain. The results of these investigations show that (effect-directed) screening processes are a suitable way of finding new interactions between foods and target molecules in the body.
3.2. Activity-Directed Fractionation
But which ingredients could be responsible for the newly discovered GABAergic activity of clove extract? There are different methodological approaches for answering this question: for example, computational chemistry methods, metabolic profile analysis, or principal component analysis. Principal component analysis is a statistical procedure by which the relationship between variables (such as the components of a food) and the assignment of a sample to a group (such as bioactive or inactive food) can be visualized.
Frequently, however, the method used to answer this question is activity-directed fractionation. In this process, the active extract is separated into multiple fractions by a chromatographic process, such as reversed-phase high-performance liquid chromatography (RP-HPLC). Each of these fractions is then tested by a bioassay. If activity is seen in one of the fractions, that one is fractionated itself—if possible, through a complementary separation technique. These steps are repeated until the activity can be assigned to a single substance.
4. Processes in the Body
4.1. Eugenol and Acetyleugenol from Clove Extract Have a Sedative Effect through the GABAA Receptor
By using activity-directed fractionation, it was possible to identify eugenol as the primary GABAergic agent in clove extracts (see Fig. 3). A second component, acetyleugenol, had significantly higher activity than eugenol itself, but its low concentration means it barely contributes to the overall activity of the extract [5].
.jpg) |
|
Figure 3. GABA active substances. |
These two neuroactive food ingredients have structural features in common with other allosteric modulators of the GABAA receptor. This also previously made it possible to show that other alkylphenols, such as carvacrol and chlorothymol, also amplify the effect of GABA on the GABAA receptor. This effect is best known for 2,6-diisopropylphenol, known as Propofol, which is used as an anesthetic in medical clinics [6,7].
4.2. Absorption and Biotransformation
It is also interesting to take a more thorough look at the processes in the body that occur when such GABAergic substances are ingested. Foods and their ingredients must first pass through the stomach and intestines, where the components are absorbed to different degrees. Subsequently, they can be biotransformed to a significant degree.
For low-molecular-weight components, it is particularly interesting to observe the activity of their metabolites. In this way, it was demonstrated that initial biotransformations take place in the intestinal tract, and the products can be more bioavailable than the original agents [8–10]. Some of the metabolites, such as pinocarveol, verbenol, and myrtenol, derivates of α- and β-pinene, can even have a stronger effect on the GABAA receptor than the original substance [7].
For foods, it is additionally necessary to take into account that they are generally a complex mixture of substances. This mixture can be made more complex by processes in the body, which can form additional substances. This poses big challenges for future studies aiming to draw conclusions about the physiological effects of such mixtures in aggregate.
4.3. Food Ingredients that Influence our Eating Behavior
Not only do foods influence brain activity but brain activity also influences what and how much we eat. It has long been known that malfunctions in certain areas of the hypothalamus, which can be caused by tumors or other things, may lead to an excessive calorie uptake. The hypothalamus is a region of the brain whose functions include regulation of hunger and appetite.
This interplay between brain function and foods can go in either direction. This means that certain foods change the activity of regions of the brain that play an important role in food consumption. The result is that these special foods are often consumed more often and in larger amounts. In this regard, even many completely healthy consumers frequently report feeling a loss of control when eating things like potato chips or chocolate. To date, little is known about the molecular causes of these phenomena.
4.4. Manganese-Enhanced Magnetic Resonance Imaging (MEMRI)
In animals, it has been possible to show the influence of food on brain activity through imaging processes. A particularly good technique for this is a variant of magnetic resonance imaging, the MEMRI technique (manganese-enhanced magnetic resonance imaging). In this process, animals are given manganese chloride as a contrast agent for up to one week. In the brain, manganese behaves like a calcium ion, which streams into every activated nerve cell as a natural signal, transmitting the information. In contrast to calcium, magnesium is not transported back out of the cell after activation, which leads to an enrichment of manganese in nerve cells that have previously been activated. The paramagnetic properties of manganese make it detectable by magnetic resonance tomography (MRT).
After a week of receiving manganese chloride, MRT makes all of the brain activity that occurred over this period visible. Because the animals can move freely during the week of the experiment, it is also possible to measure which brain regions were activated or deactivated by special foods or special food ingredients.
References
[1] J. V. Higdon, B. Frei, Coffee and Health: A Review of Recent Human Research, Crit. Rev. Food Sci. Nutr. 2006, 46, 101–123. https://doi.org/10.1080/10408390500400009
[2] B. Sicard, Die Alltagsdroge Koffein (in German), Spektrum der Wissenschaft 2003, 64–71.
[3] M. Lancel, A. Steiger, Sleep and Its Modulation by Drugs That Affect GABAA Receptor Function, Angew. Chem. Int. Ed. 1999, 111, 2852–2864. https://doi.org/10.1002/(SICI)1521-3773(19991004)38:19<2852::AID-ANIE2852>3.0.CO;2-Z
[4] S. Sahin et al., Three-Step Test System for the Identification of Novel GABAA Receptor Modulating Food Plants, Plant Foods Hum. Nutr. 2016, 71, 355–360. https://doi.org/10.1007/s11130-016-0566-1
[5] S. Sahin et al., Identification of eugenol as the major determinant of GABAA-receptor activation by aqueous Syzygium aromaticum L. (clove buds) extract, J. Funct. Foods 2017, 37, 641–649. https://doi.org/10.1016/j.jff.2017.08.033
[6] A. Kessler et al., GABAA receptor modulation by terpenoids from Sideritis extracts, Flavour Fragrance J. 2012, 27, 297–303. https://doi.org/10.1002/mnfr.201300420
[7] A. Kessler et al., GABA(A) receptor modulation by terpenoids from Sideritis extracts, Mol. Nutr. Food Res. 2014, 58, 851–862. https://doi.org/10.1002/mnfr.201300420
[8] A. Heinlein, A. Buettner, Monitoring of biotransformation of hop aroma compounds in an in vitro digestion model, Food Funct. 2012, 3, 1059–1067. https://doi.org/10.1039/C2FO30061C
[9] A. Heinlein et al., in: T. Hofmann, P. Schieberle (Eds.), 10th Wartburg Symposium on Flavor Chemistry and Biology, Deutsche Forschungsanstalt für Lebensmittelchemie, Eisenach 2014, 265–269.
[10] A. Heinlein et al., Transport of hop aroma compounds across Caco-2 monolayers, Food Funct. 2014, 5, 2719–2730. https://doi.org/10.1039/C3FO60675A
The article has been published in German as:
- Neurotrition – wie Lebensmittel auf die Gehirnfunktion wirken,
Sümeyye Sahin, Andrea Buettner, Monika Pischetsrieder,
Chem. unserer Zeit 2019, 53, 320–328.
https://doi.org/10.1002/ciuz.201900832
and was translated by Caroll Pohl-Ferry.
How Foods Affect Brain Function – Part 2
Which food components might modify brain activity patterns and induce compulsive eating?
See similar articles published on ChemistryViews.org



