Aromatic ketones are important building blocks in the synthesis of fine chemicals, including many natural products and biologically active compounds. C−H oxygenation is a highly atom-economical strategy for the synthesis of these ketones. However, peroxides are generally needed as oxidants in traditional C−H oxidation reactions, which reduces the atom economy. Recently, O2 has replaced traditional oxidants in some reactions because it is cheap, green, and readily available. However, harsh conditions or toxic solvents are frequently used, and expensive catalysts and/or ligands can also limit the practical value of these methods.
Pan Xie, Shaanxi University of Science & Technology, Xi’an, China, and colleagues have developed a visible-light-induced aerobic C−H oxidation reaction to produce aromatic ketones. The team used a photocatalytic process, with CeCl3 as the photosensitizer, polyethylene glycol 4000 (PEG 4000) as an additive, and water as the solvent under blue light and an air atmosphere. Under these conditions, they converted different diarylmethanes and aryl alkanes to the corresponding ketones.
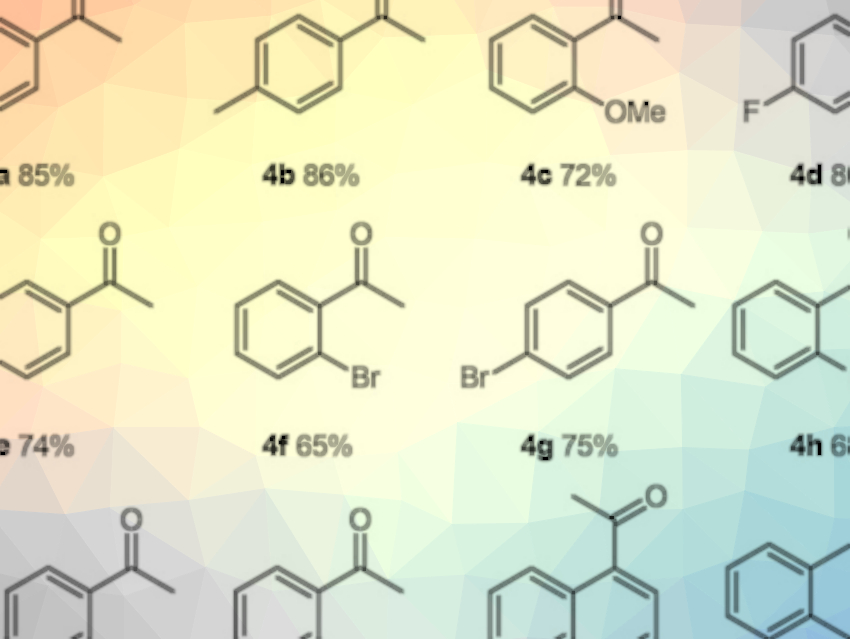
The desired aromatic ketones (examples pictured) were obtained in moderate to excellent yields. The reaction is economical, can be performed under mild conditions (i.e., at room temperature), and the use of air as an oxidant and water as the solvent makes it environmentally friendly. The method also shows good functional-group tolerance. According to the researchers, the work could provide inspiration for the development of other aqueous photochemical reactions.
- Visible‐Light‐Driven Selective Air‐Oxygenation of C−H Bond via CeCl3 Catalysis in Water,
Pan Xie, Cheng Xue, Sanshan Shi, Dongdong Du,
ChemSusChem 2021.
https://doi.org/10.1002/cssc.202100682