Electrochemical water splitting using renewable energy is a sustainable way to produce hydrogen as a fuel. However, finding suitable electrocatalysts, especially for the oxygen evolution reaction (OER), is challenging. This is due to the slow kinetics and often high overpotential of the OER. Typically, noble metal oxides are used, but their high cost prevents widespread use. Non-precious metal sulfides and oxides on a support material (e.g., a heteroatom-containing carbon material) could be useful alternatives.
Menny Shalom, Ben-Gurion University of the Negev, Beer-Sheva, Israel, and colleagues have developed an easy, scalable bottom-up synthesis of transition-metal sulfides and oxides supported on carbon-sulfur materials (pictured below). The team used low-cost metal salts (iron, nickel, or copper nitrate), pyrene, and elemental sulfur as reactants. These were mixed and heated under an N2 environment at 800 °C to obtain the desired composite materials.
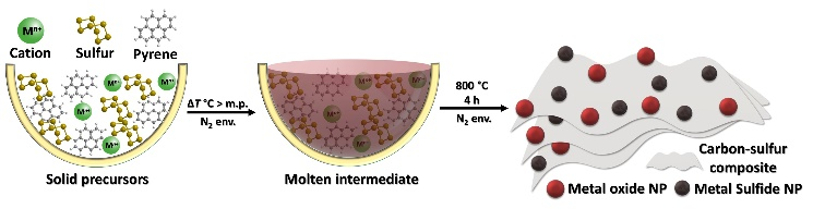
The resulting materials consist of a carbon-sulfur composite material that is homogenously decorated with nanoparticles of metal sulfide and metal oxide (pictured above on the right). The exact composition can be tuned by varying the amount of metal salt used in the synthesis. The researchers dropcast some of the prepared iron- and nickel-based composites onto a nickel foam and measured their electrochemical activity for the OER. They found that the materials exhibit state-of-the-art activity as OER electrocatalysts in alkaline media.
- Facile synthesis of carbon‐sulfur scaffold with transition‐metal sulfides and oxides as efficient electrocatalysts for oxygen evolution reaction,
Jonathan Tzadikov, Rotem Geva, Adi Azoulay, Menny Shalom,
ChemCatChem 2021.
https://doi.org/10.1002/cctc.202100572