Liquid crystals form ordered phases that have many useful applications. Timothy M. Swager, Massachusetts Institute of Technology (MIT), Cambridge, USA, and colleagues have found a way to introduce helical supramolecular structures in columnar liquid crystals by applying an electric field. Their method could be used to develop new chiroptical materials, for example, two-dimensional layers that sense chiral biomolecular assemblies, serve as organic semiconductors in device applications, or induce helicity in achiral nanostructures.
Displays and More
Liquid crystals are fascinating things. Indeed, most people probably spend longer staring at liquid crystals every day than looking out of the window. This is because liquid crystals are a key component of almost every display screen. When we watch a video on our smartphone or type words into a computer, we are relying on the ability of liquid crystals to block or open the path of polarized light in response to an electric signal.
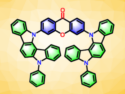
The liquid crystals used in displays are usually rod-shaped molecules with polar head groups, which arrange themselves into ordered phases along their long axis. Flat, aromatic, disk-shaped molecules can also form liquid crystal phases; however, they do it indirectly, through their supramolecular structures. Attracted by π bonding, the flat molecules assemble into columns like stacks of coins (pictured).
When these columns aggregate along their long axis, they form phases called discotic (disk-like, able to form column stacks) or columnar phases. These are two-dimensional layers with supramolecular substructures in the nanosized range. Such structurally ordered layers have applications in a wide variety of nanotechnologies.
The team introduced a chiral molecular lever that can be actuated by an electric field. Two-dimensional layers with helical substructures may have exceptional electro-optical or magnetic behavior, they say, and inducible chiroptical switches could lead to new sensory materials with chiral functionality
Seeding Conformation Changes
To obtain discotic molecules, the team prepared tetraphenylbenzene derivatives with all four phenyl rings furnished with alkyl ether moieties. They found that these molecules self-assembled into a stable liquid-crystal mesophase with hexagonal symmetry (six columnar molecules assembled as a hexagonal rosette).
In order to introduce the chiral switch, the researchers exchanged one of the four ether-substituted phenyl rings with a chiral one, containing a chiral alkyl ether chain and a polar fluorine substituent instead of the two achiral alkyl ether chains. The fluorine was intended to serve as the pivot point producing rotation in an external electric field, while the chiral ether group was expected to direct the twist.
Without an external electric field applied, the chiral molecules behaved exactly like the achiral ones. They self-assembled in similar stacks in solution, and showed the same hexagonal columnar mesophase as a thin film. The researchers then sandwiched the liquid crystals in a capacitor cell and applied a voltage. They found the mesophase increased its birefringence and showed ellipticity when analyzed by circular dichroism (CD) spectrometry. After the electric field was removed, the ellipticity slowly faded and was gone after several hours.
From Columns to Propellers in a Collective Effort
The team explained the changes by the formation of supramolecular helices. First, the electric field “pulled” on the fluorine, so the peripheral chiral phenyl groups were forced to rotate out their initial plane. With the chiral groups directing the twist, the tilt imposed on the individual molecules resulted in a propeller-like conformation of the stack as a whole.
The researchers also found that there was a collective assembly process. When they added a small amount of the chiral molecules to the achiral molecules and applied an electric field, they found that the CD signal increased rapidly. This means the few chiral molecules had transferred their helical twisting to the majority of the achiral columnar stacks.
The team has shown that phases with two-dimensional supramolecular order can be given a helical twist when placed in an electric field. The new material produced in this way may be interesting for chiro-optical switches, sensory materials, or even new chiro-magnetic applications.
- Electric-Field-Induced Chirality in Columnar Liquid Crystals,
Alberto Concellón, Ru-Qiang Lu, Kosuke Yoshinaga, Hsiu-Fu Hsu, Timothy M. Swager,
J. Am. Chem. Soc. 2021, 143, 9260–9266.
https://doi.org/10.1021/jacs.1c05268