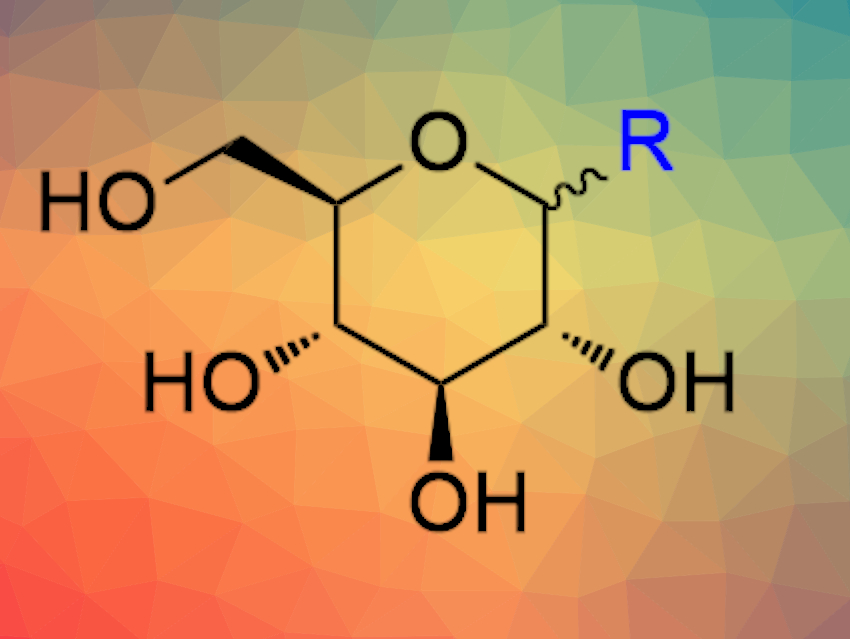
Carbohydrates can play important roles in molecular and cellular recognition events, and thus, are interesting for drug discovery. However, their use as drugs has been hampered by their relatively poor pharmacokinetic properties, e.g., high polarity. Thus, synthetic pathways towards sugar-derived molecules with improved properties and interesting biological activities would be useful.
Kamil Parkan University of Chemistry and Technology Prague, Czech Republic, and colleagues have developed a synthesis of a range of aryl-C-glycosides—promising glycomimetic molecules with drug-like properties. The team used a silicon-based synthon, 1-diisopropylsilyl-D-glucal (pictured below on the left). This approach enables the convenient introduction of a range of aromatic substituents into a sugar-derived molecule via a mild Pd-catalysed Hiyama cross-coupling reaction without the need for protecting groups. [PdCl(allyl)]2 was used as a catalyst, tetramethylammonium fluoride tetrahydrate (TMAF·4H2O) as a fluoride source, and N,N-dimethylformamide (DMF) as the solvent.
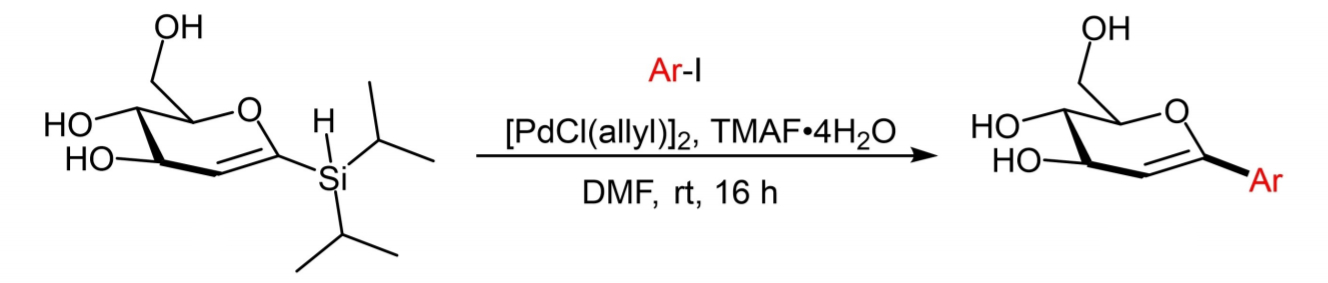
The resulting unprotected pseudo-C-glucosides can be stereoselectively converted into the corresponding α- and β-C-glucosides, as well as 2-deoxy-β-C-glucosides. Using this approach, the team developed a short and efficient synthesis of dapagliflozin, a commercial drug used for the treatment of diabetes, as well as its analogues with 2-deoxy-β-D-gluco and α-D-gluco configuration.
- Facile Approach to C-Glucosides by Using a Protecting-Group-Free Hiyama Cross-Coupling Reaction: High-Yielding Dapagliflozin Synthesis,
Karolína Vaňková, Michal Rahm, Jan Choutka, Radek Pohl, Kamil Parkan,
Chem. Eur. J. 2021.
https://doi.org/10.1002/chem.202101052