Arne Wilhelm Kaurin Tiselius was born on August 10, 1902, in Stockholm, Sweden. He studied chemistry at Uppsala University, Sweden, and then worked as a Research Assistant at Theodor (The) Svedberg’s laboratory in Uppsala. He obtained his Ph.D. there in 1930 and became an Assistant Professor. He was promoted to Research Professor of Biochemistry in Uppsala in 1938, a post he held for 30 years. Arne Tiselius died on October 29, 1971, in Uppsala.
Tiselius is best known for developing electrophoresis as a method for molecular separation. The technique is based on molecules migrating along an applied electric field through a stationary medium. Depending on their charge and size, the molecules migrate at different speeds. This can be used to separate different substances from one another and serves as an important tool in chemical analysis, especially in biochemistry.
Tiselius developed “moving-boundary electrophoresis” and used it for the separation of mixtures of proteins [1]. This approach uses a cell filled with a buffer solution, with electrodes immersed at its ends. On applying a voltage, the compounds in an applied sample will migrate to the anode or the cathode, depending on their charges. The boundaries between the separated compounds can then be detected, e.g., via changes in the refractive index of the solution. Using this method, Tiselius separated the main protein components of blood serum, i.e., albumin and globulins. Later, filter paper was used as a medium for electrophoresis.
The Nobel Prize in Chemistry 1948 was awarded to Tiselius “for his research on electrophoresis and adsorption analysis, especially for his discoveries concerning the complex nature of the serum proteins.” He also worked on measuring the diffusion of water vapor and other gases into zeolite crystals.
Arne Tiselius is the answer to Guess the Chemist (118).
References
- [1] A new apparatus for electrophoretic analysis of colloidal mixtures,
A. Tiselius,
Trans. Faraday Soc. 1937, 33, 524.
https://doi.org/10.1039/tf9373300524
Sources
- Obituary: Arne Tiselius, 1902 – 1971,
R. A. Kekwick, K. O. Pedersen
Biogr. Mem. Fellows R. Soc. 1974, 20, 400–428.
https://doi.org/10.1098/rsbm.1974.0018
Selected Publications
- A New Method for the Determination of the Mobility of Proteins,
T. Svedberg, A. Tiselius,
J. Am. Chem. Soc. 1926, 48, 2272–2278.
https://doi.org/10.1021/ja01420a004 - Diffusion of Water in a Zeolite Crystal,
A. Tiselius,
Nature 1934, 133, 212–213.
https://doi.org/10.1038/133212a0 - Adsorption and Diffusion in Zeolite Crystals.,
A. Tiselius,
J. Phys. Chem. 1936, 40, 223–232.
https://doi.org/10.1021/j150371a008 - Electrophoresis of serum globulin,
A. Tiselius,
Biochem. J. 1937, 31, 1464–1477.
https://doi.org/10.1042/bj0311464 - Elektrophorese von Eiweiss in Filtrierpapier,
A. Tiselius, H. D. Cremer,
Biochem Z. 1950, 320, 273–283. - Reflections from Both Sides of the Counter,
A. Tiselius,
Annu. Rev. Biochem. 1968, 37, 1–23.
https://doi.org/10.1146/annurev.bi.37.070168.000245
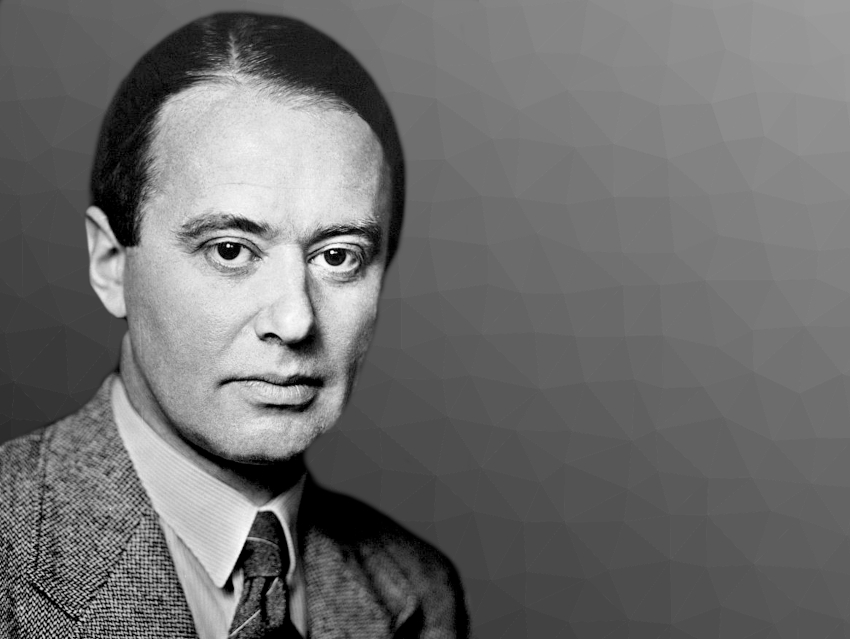


The basis of separation is not the molecule’s charge but the charge to mass ratio resp. charge to molecular volume ratio. Else molecules with the same charge would not separate.
Dear Reader, Thank you very much for pointing this out. We have modified the text accordingly. Your ChemistryViews Team